MALVERN, Pa. and PHILADELPHIA, May 21, 2012 /PRNewswire/ --Auxilium Pharmaceuticals, Inc. (AUXL) and GlaxoSmithKline LLC, the U.S. subsidiary of GlaxoSmithKline plc (GSK.L), announced today that they have entered into an agreement for the co-promotion of Testim 1% (testosterone gel), which is indicated for testosterone replacement therapy in adult males for conditions associated with a deficiency or absence of testosterone.
(Logo: http://photos.prnewswire.com/prnh/20101202/MM10881LOGO )
"Today, GlaxoSmithKline and Auxilium have forged a collaboration to expand our reach to U.S. physicians who treat men with low testosterone and its resulting symptoms, known as hypogonadism, which we believe is a prevalent, but poorly recognized condition," said Adrian Adams, Chief Executive Officer and President of Auxilium. "While the Auxilium sales force has consistently performed well in an increasingly competitive environment, adding the strength and breadth of GSK's U.S. commercialization organization will further enhance our sales efforts with U.S. physicians who treat this condition."
"Urology remains one of GSK's key therapeutic areas, and our sales professionals are well positioned to provide physicians with information on the treatment of hypogonadism," said Cheryl MacDiarmid, Vice President of GSK's Cardiovascular/Metabolic/Urology business unit. "We look forward to our collaboration with Auxilium and adding Testim to our portfolio so we can help meet the needs of men with this condition."
Under the terms of the agreement, Auxilium granted GSK the exclusive right to co-promote the sale of Testim with Auxilium in the U.S. through September 30, 2015. GSK will promote Testim using a sizeable established field sales force which has relationships with current Testosterone Replacement Therapy (TRT) prescribers, particularly primary care physicians, in the U.S. These GSK sales representatives currently promote a range of cardiovascular, metabolic and urology products, and Testim will complement GSK's existing portfolio of products.
Auxilium and GSK have agreed on a baseline revenue forecast for Testim through September 30, 2015, and GSK will be compensated to the extent that Testim net sales exceed this baseline. In addition, in certain circumstances, Auxilium will pay GSK specified tail payments following the term of the agreement. The GSK sales force is expected to begin promoting Testim to physicians early in the third quarter 2012. Auxilium will remain responsible for all Testim commercial drug manufacturing, supply, and regulatory activities.
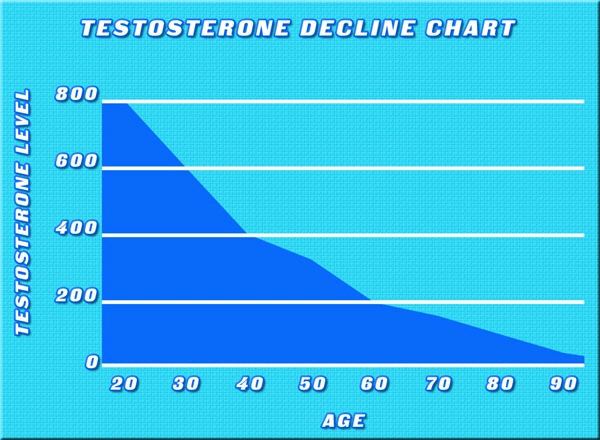
About Low Testosterone Testosterone and dihydrotestosterone (DHT), endogenous androgens, are responsible for normal growth and development of the male sex organs and for maintenance of secondary sex characteristics. Male hypogonadism results from insufficient secretion of testosterone and is characterized by low serum testosterone concentrations (total testosterone levels below 300 ng / dL). Symptoms associated with male hypogonadism include decreased sexual desire with or without impotence, fatigue and loss of energy, mood depression, regression of secondary sexual characteristics, and osteoporosis. Hypogonadism is a risk factor for osteoporosis in men.(1)
About Testim Testim is a proprietary, clear, topical gel containing 1% testosterone, the same type of hormone that is produced by the human body. When applied once-daily to the upper arms and shoulders, clinical studies have shown that Testim helps restore and maintain testosterone levels. Once Testim is absorbed through the skin, it enters the bloodstream and helps return testosterone to normal levels.
According to IMS data for 2011, U.S. sales of testosterone therapies have grown to more than $1.6 billion per year, with sales of testosterone gels in the U.S. comprising $1.4 billion.
Important Safety Information Testim has not been clinically evaluated in males under 18 years of age. Virilization has been reported in children who were secondarily exposed to testosterone gel. Children should avoid contact with unwashed or unclothed application sites in men using testosterone gel. Healthcare providers should advise patients to strictly adhere to recommended instructions for use.
Excerpt from:
Auxilium Pharmaceuticals, Inc. and GlaxoSmithKline LLC Enter Into a Co-Promotion Agreement for Testim® in the U.S.
Contact Us Today For A Free Consultation

- Part 2: Effect of Testosterone Replacement Therapy on Prostate Tissue in Men with Late-Onset Hypogonadism (Dramatic Health) [Last Updated On: March 24th, 2018] [Originally Added On: May 7th, 2011]
- Full: Effect of Testosterone Replacement Therapy on Prostate Tissue in Men with Late-Onset Hypogonadism (Dramatic Health) [Last Updated On: May 3rd, 2023] [Originally Added On: May 7th, 2011]
- Testosterone Replacement Therapy (TRT): Optimizing Clinical Outcomes - Michael Aziz, MD [Last Updated On: November 12th, 2023] [Originally Added On: May 8th, 2011]
- Part 1: Effect of Testosterone Replacement Therapy on Prostate Tissue in Men with Late-Onset Hypogonadism (Dramatic Health) [Last Updated On: March 24th, 2018] [Originally Added On: May 8th, 2011]
- Part 4: Effect of Testosterone Replacement Therapy on Prostate Tissue in Men with Late-Onset Hypogonadism (Dramatic Health) [Last Updated On: November 12th, 2023] [Originally Added On: May 30th, 2011]
- Part 3: Effect of Testosterone Replacement Therapy on Prostate Tissue in Men with Late-Onset Hypogonadism (Dramatic Health) [Last Updated On: November 15th, 2023] [Originally Added On: June 2nd, 2011]
- Testosterone Replacement [Last Updated On: November 23rd, 2023] [Originally Added On: June 7th, 2011]
- Medical Professor on Testosterone Replacement Therapy [Last Updated On: November 17th, 2023] [Originally Added On: June 14th, 2011]
- Sean McCorkle Discusses Testosterone Replacement Therapy [Last Updated On: November 18th, 2023] [Originally Added On: July 11th, 2011]
- Use of Testosterone in Men With Prostate Cancer [Last Updated On: November 22nd, 2023] [Originally Added On: September 28th, 2011]
- Low Testosterone (Low T) - Video [Last Updated On: November 25th, 2024] [Originally Added On: December 10th, 2011]
- Transdermal Drug Delivery - Technologies, Markets, and Companies [Last Updated On: April 29th, 2025] [Originally Added On: February 2nd, 2012]
- How Testosterone Replacement Therapy Builds Muscle and Stops Pain [Last Updated On: February 4th, 2024] [Originally Added On: February 4th, 2012]
- Teva, BioSante’s Testosterone Gel for Men Wins Approval From U.S. FDA [Last Updated On: January 11th, 2018] [Originally Added On: February 15th, 2012]
- FDA approves Teva, BioSante testosterone gel [Last Updated On: January 18th, 2018] [Originally Added On: February 15th, 2012]
- Teva Fourth-Quarter Profit Rises on Cephalon Purchase [Last Updated On: January 16th, 2018] [Originally Added On: February 15th, 2012]
- Renowned Dr. Oz and the Acclaimed Financial Times Now Have Featured Dr. Lionel Bisson, Founder of ... [Last Updated On: April 27th, 2025] [Originally Added On: February 17th, 2012]
- Omaha man says testosterone replacement therapy changed his life [Last Updated On: December 15th, 2017] [Originally Added On: May 5th, 2012]
- Hormone therapy results in weight loss [Last Updated On: January 27th, 2018] [Originally Added On: June 25th, 2012]
- Testosterone in Women-Putting Your Sex Drive Bacl On Track - Video [Last Updated On: December 31st, 2024] [Originally Added On: November 2nd, 2012]
- Men's Health PITCH: Testosterone - Video [Last Updated On: January 1st, 2025] [Originally Added On: November 2nd, 2012]
- Dr. Karron Power Appears on Nightline - Testosterone Therapy - Video [Last Updated On: January 1st, 2025] [Originally Added On: November 2nd, 2012]
- Testosterone Replacement Therapy: Who is TRT Best For? - Video [Last Updated On: January 2nd, 2025] [Originally Added On: November 2nd, 2012]
- Female sex-enhancing nasal spray undergoing clinical trials [Last Updated On: October 24th, 2015] [Originally Added On: November 2nd, 2012]
- Andropause: A Diagnosis Whose Time Has Come [Last Updated On: June 3rd, 2025] [Originally Added On: November 16th, 2012]
- Dealing With Mood Disorders During the Holidays (Depression, Anxiety, Depersonalization) - Video [Last Updated On: February 7th, 2025] [Originally Added On: November 26th, 2012]
- Testosterone Roundtable -- Hypergonadism and Testosterone Replacement Therapy (Part 6) - Video [Last Updated On: February 19th, 2025] [Originally Added On: December 10th, 2012]
- Battling my Testosterone Replacement Therapy Doctor, Carpal Tunnel Syndrome, and Weightlifting - Video [Last Updated On: March 12th, 2013] [Originally Added On: March 12th, 2013]
- Low T? Testosterone Replacement Therapy - Video [Last Updated On: March 13th, 2013] [Originally Added On: March 13th, 2013]
- Testosterone Replacement Therapy: Nothing To Be Ashamed Of: Strike First Nutrition - Video [Last Updated On: May 18th, 2013] [Originally Added On: May 18th, 2013]
- Testosterone Replacement Therapy: Symptoms of Low Testosterone - Strike First Nutrition - Video [Last Updated On: May 18th, 2013] [Originally Added On: May 18th, 2013]
- Testosterone Replacement Therapy Testimonial - Video [Last Updated On: June 15th, 2013] [Originally Added On: June 15th, 2013]
- Taurus Male Clinic Testosterone Replacement Therapy - Video [Last Updated On: July 28th, 2013] [Originally Added On: July 28th, 2013]
- Can I Quit TRT Or Testosterone Replacement Therapy? By Low Testosterone Expert Dr David Asher - Video [Last Updated On: March 16th, 2017] [Originally Added On: August 7th, 2013]
- What Are The Side Effects of TRT Testosterone Replacement Therapy? By Low T Expert Dr. David Asher - Video [Last Updated On: August 7th, 2013] [Originally Added On: August 7th, 2013]
- Bill Jones 1513 Testosterone Replacement Therapy Testimonial - Video [Last Updated On: August 8th, 2013] [Originally Added On: August 8th, 2013]
- 2013-08-06 Testosterone Replacement Therapy - Video [Last Updated On: August 10th, 2013] [Originally Added On: August 10th, 2013]
- Testosterone Replacement Therapy West Palm Beach Florida - Video [Last Updated On: August 18th, 2013] [Originally Added On: August 18th, 2013]
- Testosterone Replacement Therapy Testimony from Patient of Body Renew Medical in Lees Summit MO - Video [Last Updated On: September 2nd, 2013] [Originally Added On: September 2nd, 2013]
- FGSW - An Update On My Testosterone Replacement Therapy: Doctor's Visit 09.16.13 - Video [Last Updated On: September 19th, 2013] [Originally Added On: September 19th, 2013]
- Testosterone Replacement Therapy - Testosterone Treatment [Last Updated On: March 25th, 2025] [Originally Added On: October 31st, 2013]
- Testosterone Side Effects from Testosterone Replacement Therapy [Last Updated On: January 11th, 2018] [Originally Added On: November 3rd, 2013]
- FGSW - An Update On My Testosterone Replacement Therapy (TRT) 11.08.13: Getting No Sleep! - Video [Last Updated On: November 14th, 2013] [Originally Added On: November 14th, 2013]
- WebMD: Erectile Dysfunction: Testosterone Replacement Therapy [Last Updated On: December 6th, 2017] [Originally Added On: November 25th, 2013]
- Testosterone Replacement Therapy! Male Hormones! [Last Updated On: January 15th, 2018] [Originally Added On: November 25th, 2013]
- FGSW - Testosterone Replacement Therapy TRT Update: Am I Back Where I Started - 11.25.13 - Video [Last Updated On: November 27th, 2013] [Originally Added On: November 27th, 2013]
- The benefits and risks of testosterone replacement therapy: a ... [Last Updated On: January 12th, 2018] [Originally Added On: December 8th, 2013]
- Testosterone replacement therapy can carry health risks - CBS News [Last Updated On: January 16th, 2018] [Originally Added On: December 8th, 2013]
- Reclaim Your Energy and Sex Drive | Testosterone Replacement ... [Last Updated On: April 6th, 2025] [Originally Added On: December 21st, 2013]
- Low Testosterone Therapy and Treatment - Do You Have Low ... [Last Updated On: October 24th, 2015] [Originally Added On: December 24th, 2013]
- How Long Does it Take for Testosterone Replacement Therapy to ... [Last Updated On: December 30th, 2017] [Originally Added On: January 3rd, 2014]
- Testosterone Therapy - Bioidentical Testosterone Replacement [Last Updated On: January 23rd, 2018] [Originally Added On: January 9th, 2014]
- Transdermal testosterone replacement therapy - Video abstract 43475 - Video [Last Updated On: January 11th, 2014] [Originally Added On: January 11th, 2014]
- Testosterone Therapy - Bioidentical Testosterone Replacement ... [Last Updated On: December 21st, 2017] [Originally Added On: January 13th, 2014]
- Testosterone Side Effects from Testosterone Replacement ... [Last Updated On: December 8th, 2017] [Originally Added On: January 20th, 2014]
- MyAntiAgingMD The Leader In Testosterone Replacement Therapy - Video [Last Updated On: October 21st, 2020] [Originally Added On: January 29th, 2014]
- The benefits and risks of testosterone replacement therapy ... [Last Updated On: October 15th, 2020] [Originally Added On: February 6th, 2014]
- The Secret Female Hormone: How Testosterone Replacement Can Change Your Life [Last Updated On: October 28th, 2020] [Originally Added On: February 11th, 2014]
- Testosterone Replacement Therapy found to be linked to Heart Problems [Last Updated On: April 19th, 2025] [Originally Added On: February 25th, 2014]
- Serious Side Effects Linked to Testosterone Therapy [Last Updated On: September 17th, 2025] [Originally Added On: February 27th, 2014]
- Doctor Reveals How Getting off Testosterone Will Hurt Fighters on It [Last Updated On: November 27th, 2020] [Originally Added On: March 4th, 2014]
- California joins Nevada in banning testosterone replacement therapy [Last Updated On: October 4th, 2020] [Originally Added On: March 6th, 2014]
- Testosterone Replacement Therapy: How to Administer Expert TRT By John K. Crisler, DO - Video [Last Updated On: October 31st, 2020] [Originally Added On: March 9th, 2014]
- FGSW - Week 14 Weigh-In & Update: Going Back To The Endocrinologist - Video [Last Updated On: March 29th, 2014] [Originally Added On: March 29th, 2014]
- Drug Recall Attorney at Herrera Law Firm, Inc., Comments on Reported Link Between Testosterone Drugs and Heart Attack ... [Last Updated On: November 30th, 2020] [Originally Added On: April 2nd, 2014]
- FGSW - Testosterone Replacement Therapy Update 03.27.14: An Improvement?? - Video [Last Updated On: October 8th, 2020] [Originally Added On: April 2nd, 2014]
- FGSW Weekly Weigh In & Update 15 Still Working On Mobility 03 31 14 - Video [Last Updated On: October 15th, 2020] [Originally Added On: April 5th, 2014]
- Discover the risk of prostate cancer with testosterone replacement therapy - Video [Last Updated On: October 1st, 2020] [Originally Added On: April 5th, 2014]
- FGSW - Weekly Weigh In & Update 15: Still Working On Mobility 03.31.14 - Video [Last Updated On: October 3rd, 2020] [Originally Added On: April 7th, 2014]
- Testosterone Replacement Therapy - Video [Last Updated On: October 1st, 2020] [Originally Added On: April 12th, 2014]
- Reclaim Your Energy and Sex Drive | Testosterone ... [Last Updated On: April 21st, 2025] [Originally Added On: April 14th, 2014]
- Low Testosterone Claims: Options in a Growing Class Action Suit - Video [Last Updated On: November 25th, 2020] [Originally Added On: April 26th, 2014]
- Acrux falls 15% on sales warning [Last Updated On: September 28th, 2025] [Originally Added On: April 28th, 2014]
- Testosterone Replacement Therapy in Men - myVMC [Last Updated On: November 1st, 2020] [Originally Added On: April 30th, 2014]
- NJ-Based Law Firm Exploring Potential Legal Claims from Side Effects of Testosterone Replacement Therapy [Last Updated On: October 22nd, 2020] [Originally Added On: May 1st, 2014]
- Viewer Mail - Testosterone Replacement Therapy/TRT Not Natty, Balls Busted, etc. - Video [Last Updated On: October 2nd, 2020] [Originally Added On: May 4th, 2014]
- Seen At 11: Experts Urge Caution When Using Popular Hormone Replacement Therapy [Last Updated On: November 11th, 2020] [Originally Added On: May 10th, 2014]
- Are Low-T Drugs Putting Patients At High Risk? [Last Updated On: October 15th, 2020] [Originally Added On: May 10th, 2014]
- Seen At 11: Testosterone Replacement Therapy Could Come With Serious Side Effects - Video [Last Updated On: May 11th, 2014] [Originally Added On: May 11th, 2014]
- 1513 Hormone Replacement Therapy for Men - Video [Last Updated On: October 15th, 2020] [Originally Added On: June 13th, 2014]
Word Count: 626