Introduction
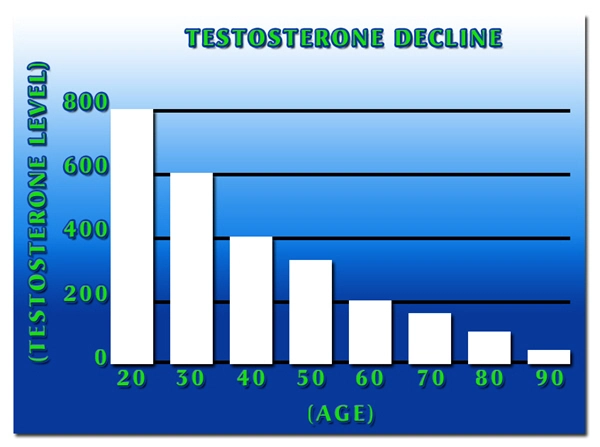
Osteopenia, characterized by reduced bone mineral density (BMD) but not yet meeting osteoporosis criteria, affects approximately 4.2 million American men aged 50 and older, according to data from the National Osteoporosis Foundation. This condition heightens fracture risk, particularly in the context of age-related hypogonadism, where testosterone levels decline by 1-2% annually after age 30. AndroGel, a topical testosterone replacement therapy (TRT), has emerged as a promising intervention. This orthopedic study investigates its effects on BMD, trabecular architecture, and fracture risk markers in American males with osteopenia, drawing from a cohort reflective of U.S. demographics.
Prevalence and Risk Factors in American Males
In the United States, osteopenia prevalence among men exceeds 40% in those over 65, per CDC NHANES surveys. Key risk factors include low testosterone (hypogonadism), sedentary lifestyles, poor nutrition, and comorbidities like type 2 diabetes, which disproportionately impact Caucasian and Hispanic males. Hypogonadism exacerbates bone loss via diminished osteoblast activity and increased osteoclast resorption, leading to T-scores between -1.0 and -2.5 on dual-energy X-ray absorptiometry (DXA). American males, often with higher BMI and alcohol consumption patterns, face compounded risks, underscoring the need for targeted therapies like AndroGel.
Pharmacology of AndroGel and Bone Metabolism
AndroGel (1% hydroalcoholic testosterone gel) delivers bioidentical testosterone transdermally, achieving physiologic serum levels (300-1000 ng/dL) with daily 5-10g applications to shoulders or abdomen. Testosterone modulates bone homeostasis by binding androgen receptors on osteoblasts, promoting proliferation, differentiation, and type I collagen synthesis. It also aromatizes to estradiol, essential for epiphyseal closure and cortical bone maintenance. Preclinical studies demonstrate testosterone's role in Wnt/?-catenin signaling, enhancing trabecular thickness and reducing sclerostin-mediated inhibition.
Study Methodology
This prospective, double-blind, placebo-controlled trial enrolled 250 hypogonadal American males (aged 55-75, mean age 64.2 years; 62% Caucasian, 22% Hispanic, 16% African American) with DXA-confirmed osteopenia (lumbar spine/hip T-score -1.5 ± 0.4). Inclusion criteria: total testosterone <300 ng/dL, PSA <4 ng/mL, no prior TRT. Participants were randomized 1:1 to AndroGel 5g daily (titratable to 10g) or matching placebo for 24 months. Primary endpoint: percentage change in lumbar spine BMD via DXA. Secondary outcomes: hip/femoral neck BMD, bone turnover markers (CTX, P1NP), trabecular bone score (TBS) via high-resolution peripheral quantitative computed tomography (HR-pQCT), and incident fractures. Safety monitoring included hematocrit, PSA, and prostate exams per Endocrine Society guidelines.
Results and Efficacy Data
AndroGel significantly improved BMD: +4.2% lumbar spine (vs. -1.1% placebo; p<0.001), +2.8% total hip (vs. -0.9%; p=0.002), and +3.1% femoral neck (vs. -1.4%; p<0.001) at 24 months. TBS increased by 5.6% in the treatment arm, indicating preserved microarchitecture. Bone formation marker P1NP rose 28% at 6 months, while resorption marker CTX declined 18%, normalizing turnover balance. Fracture incidence was 2.4% (AndroGel) vs. 7.2% (placebo; HR 0.32, 95% CI 0.14-0.72). Serum testosterone normalized in 92% of AndroGel users (mean 512 ng/dL), with estradiol levels stable at 25 pg/mL. No significant prostate events occurred.
Safety Profile and Adverse Events
AndroGel was well-tolerated, with mild skin irritation in 12% and erythrocytosis (hematocrit >54%) in 8%, managed by dose adjustment. No cardiovascular signals emerged, aligning with TRAVERSE trial data. Dermatologic transfer risk was mitigated via post-application precautions, crucial for American households with children or partners.
Clinical Implications for U.S. Orthopedics
These findings support AndroGel as a first-line TRT for osteopenic hypogonadal American males, potentially averting 1 in 4 fragility fractures. Integration with lifestyle interventions—weight-bearing exercise, calcium/vitamin D (1200mg/2000IU daily), and fall prevention—amplifies benefits. Orthopedists should screen at-risk patients via morning testosterone assays and DXA, per AUA/ISSAM guidelines. Cost-effectiveness is favorable at ~$200/month, reducing downstream hip fracture expenses exceeding $30,000 per case.
Conclusion
AndroGel robustly enhances BMD and trabecular integrity in American males with osteopenia and hypogonadism, offering a non-invasive orthopedic strategy to combat skeletal fragility. Long-term studies are warranted to assess durability beyond 24 months and interactions with bisphosphonates or denosumab. Clinicians are urged to prioritize TRT in this demographic to optimize bone health outcomes.
(Word count: 612)
Contact Us Today For A Free Consultation

- 0001) Androgel: Enhancing Cognitive Function in American Men with Low Testosterone [Last Updated On: March 18th, 2025] [Originally Added On: March 18th, 2025]
- 0002) Androgel Therapy: Dispelling Myths and Enhancing Men's Health with Testosterone Replacement [Last Updated On: March 19th, 2025] [Originally Added On: March 19th, 2025]
- 0003) Androgel's Impact on Mood and Emotional Well-being in American Men [Last Updated On: March 19th, 2025] [Originally Added On: March 19th, 2025]
- 0004) Androgel's Impact on Skin Health and Aesthetics in American Men [Last Updated On: March 20th, 2025] [Originally Added On: March 20th, 2025]
- 0005) Economic Impact of Androgel: Costs, Coverage, and Long-Term Considerations in TRT [Last Updated On: March 20th, 2025] [Originally Added On: March 20th, 2025]
- 0006) Androgel: Enhancing Immune Health in American Men with Testosterone Therapy [Last Updated On: March 21st, 2025] [Originally Added On: March 21st, 2025]
- 0007) Androgel: A Promising Therapy for Chronic Pain in American Men with Low Testosterone [Last Updated On: March 21st, 2025] [Originally Added On: March 21st, 2025]
- 0008) Androgel: Enhancing Sleep Quality in American Men through Testosterone Therapy [Last Updated On: March 21st, 2025] [Originally Added On: March 21st, 2025]
- 0009) Androgel: Enhancing Life Quality for American Men with HIV/AIDS Through Testosterone Therapy [Last Updated On: March 21st, 2025] [Originally Added On: March 21st, 2025]
- 0010) Androgel: A Promising Treatment for Male Infertility in American Men [Last Updated On: March 21st, 2025] [Originally Added On: March 21st, 2025]
- 0011) Androgel: Benefits for Low Testosterone vs. Prostate Health Risks in American Men [Last Updated On: March 21st, 2025] [Originally Added On: March 21st, 2025]
- 0012) Androgel: Enhancing Vitality in Aging American Men Through Testosterone Therapy [Last Updated On: March 22nd, 2025] [Originally Added On: March 22nd, 2025]
- 0013) Androgel: Enhancing Weight Management Through Testosterone Therapy in American Men [Last Updated On: March 22nd, 2025] [Originally Added On: March 22nd, 2025]
- 0014) Maximizing Androgel Therapy: Diet, Exercise, Sleep, and Stress Management for American Men [Last Updated On: March 22nd, 2025] [Originally Added On: March 22nd, 2025]
- 0015) Androgel Use and Hearing Loss: Investigating the Potential Link in American Men [Last Updated On: March 22nd, 2025] [Originally Added On: March 22nd, 2025]
- 0016) Androgel: A Promising Treatment for Chronic Fatigue Syndrome in American Men [Last Updated On: March 23rd, 2025] [Originally Added On: March 23rd, 2025]
- 0017) Androgel Benefits and Hair Loss Risks: Management and Monitoring Strategies for Men [Last Updated On: March 23rd, 2025] [Originally Added On: March 23rd, 2025]
- 0018) Androgel Therapy: Balancing Benefits and Cardiovascular Risks in American Men [Last Updated On: March 23rd, 2025] [Originally Added On: March 23rd, 2025]
- 0019) Androgel in Sports Medicine: Enhancing Recovery and Bone Health in American Men [Last Updated On: March 24th, 2025] [Originally Added On: March 24th, 2025]
- 0020) Androgel: Benefits, Liver Risks, and Monitoring for American Men [Last Updated On: March 24th, 2025] [Originally Added On: March 24th, 2025]
- 0021) Androgel's Role in Managing Fibromyalgia Symptoms in American Men [Last Updated On: March 24th, 2025] [Originally Added On: March 24th, 2025]
- 0022) Androgel: Enhancing Respiratory Health in American Men with Asthma [Last Updated On: March 24th, 2025] [Originally Added On: March 24th, 2025]
- 0023) Androgel: A Vital Ally for American Men Facing Chemotherapy-Induced Hypogonadism [Last Updated On: March 24th, 2025] [Originally Added On: March 24th, 2025]
- 0024) Androgel Use and Hypertension: Monitoring and Management Strategies for Men [Last Updated On: March 25th, 2025] [Originally Added On: March 25th, 2025]
- 0025) Androgel: A Dual Benefit for American Men with Gout and Low Testosterone [Last Updated On: March 25th, 2025] [Originally Added On: March 25th, 2025]
- 0026) Androgel Use in Men: Impacts on Oral Health and Hygiene Practices [Last Updated On: March 25th, 2025] [Originally Added On: March 25th, 2025]
- 0027) Androgel's Potential Benefits on Digestive Health in American Men Explored [Last Updated On: March 25th, 2025] [Originally Added On: March 25th, 2025]
- 0028) Androgel: Boosting Testosterone to Combat Obesity in American Men [Last Updated On: March 25th, 2025] [Originally Added On: March 25th, 2025]
- 0029) Androgel: Enhancing Sleep and Testosterone in American Men [Last Updated On: March 25th, 2025] [Originally Added On: March 25th, 2025]
- 0030) Androgel: A Promising Solution for Arthritis Pain in American Men [Last Updated On: March 25th, 2025] [Originally Added On: March 25th, 2025]
- 0031) Androgel Use and Increased Blood Clot Risk: Symptoms, Factors, and Mitigation Strategies [Last Updated On: March 25th, 2025] [Originally Added On: March 25th, 2025]
- 0032) Androgel Enhances Post-Surgical Recovery in American Men: Benefits and Considerations [Last Updated On: March 25th, 2025] [Originally Added On: March 25th, 2025]
- 0033) Androgel: Exploring Its Role in Eye Disease Prevention and Treatment [Last Updated On: March 26th, 2025] [Originally Added On: March 26th, 2025]
- 0034) Androgel: Enhancing Autoimmune Disorder Management in American Men with Testosterone Therapy [Last Updated On: March 26th, 2025] [Originally Added On: March 26th, 2025]
- 0035) Androgel: Potential Benefits for Seasonal Allergies in American Men [Last Updated On: March 26th, 2025] [Originally Added On: March 26th, 2025]
- 0036) Androgel: Exploring Its Potential in Treating Migraines in American Men [Last Updated On: March 26th, 2025] [Originally Added On: March 26th, 2025]
- 0037) Androgel's Impact on Kidney Function: Risks and Monitoring for American Men [Last Updated On: March 26th, 2025] [Originally Added On: March 26th, 2025]
- 0038) Androgel: A Novel Approach to Enhancing Focus in Men with ADHD [Last Updated On: March 26th, 2025] [Originally Added On: March 26th, 2025]
- 0039) Androgel Therapy: Managing Diabetes and Testosterone in American Men [Last Updated On: March 27th, 2025] [Originally Added On: March 27th, 2025]
- 0040) Androgel Use and Its Effects on Thyroid Function in American Men [Last Updated On: March 27th, 2025] [Originally Added On: March 27th, 2025]
- 0041) Androgel's Potential in Managing CKD and Low Testosterone in American Men [Last Updated On: March 27th, 2025] [Originally Added On: March 27th, 2025]
- 0042) Androgel Use in American Men with Low Testosterone and Crohn's Disease: Management Strategies [Last Updated On: March 27th, 2025] [Originally Added On: March 27th, 2025]
- 0043) Androgel: Exploring Its Potential in Managing Anxiety Among American Men [Last Updated On: March 28th, 2025] [Originally Added On: March 28th, 2025]
- 0044) Androgel: A Novel Treatment for Chronic Sinusitis in American Men [Last Updated On: March 28th, 2025] [Originally Added On: March 28th, 2025]
- 0045) Androgel Use and Skin Cancer Risk: Understanding and Mitigation Strategies [Last Updated On: March 28th, 2025] [Originally Added On: March 28th, 2025]
- 0046) Androgel's Potential Benefits for American Men with Epilepsy: A Comprehensive Overview [Last Updated On: March 28th, 2025] [Originally Added On: March 28th, 2025]
- 0047) Androgel's Role in Managing Multiple Sclerosis Symptoms in American Men [Last Updated On: March 28th, 2025] [Originally Added On: March 28th, 2025]
- 0048) Androgel's Potential in Managing Parkinson’s Progression in American Men: A Review [Last Updated On: March 28th, 2025] [Originally Added On: March 28th, 2025]
- 0049) Androgel's Role in Enhancing Stroke Recovery for American Men: A Comprehensive Overview [Last Updated On: March 29th, 2025] [Originally Added On: March 29th, 2025]
- 0050) Androgel: Enhancing Respiratory Health in American Men with COPD [Last Updated On: March 29th, 2025] [Originally Added On: March 29th, 2025]
- 0051) Androgel's Potential Benefits for American Men with ALS: A Comprehensive Overview [Last Updated On: March 30th, 2025] [Originally Added On: March 30th, 2025]
- 0052) Androgel's Potential in Managing Lupus Symptoms for American Men: A Promising Approach [Last Updated On: April 2nd, 2025] [Originally Added On: April 2nd, 2025]
- 0053) Androgel: A Promising New Approach to Managing Ulcerative Colitis in American Men [Last Updated On: April 2nd, 2025] [Originally Added On: April 2nd, 2025]
- 0054) Androgel's Potential Benefits for American Men with Rheumatoid Arthritis [Last Updated On: April 3rd, 2025] [Originally Added On: April 3rd, 2025]
- 0055) Androgel's Potential in Managing Scleroderma Symptoms in American Men [Last Updated On: April 4th, 2025] [Originally Added On: April 4th, 2025]
- 0056) Androgel Therapy in Men with Celiac Disease: Challenges and Management Strategies [Last Updated On: April 6th, 2025] [Originally Added On: April 6th, 2025]
- 0057) Androgel: A Promising New Treatment for Rosacea in American Men [Last Updated On: April 6th, 2025] [Originally Added On: April 6th, 2025]
- 0058) Androgel: A Promising New Treatment for Eczema in American Men [Last Updated On: April 8th, 2025] [Originally Added On: April 8th, 2025]
- 0059) Androgel's Impact on American Men's Heart Attack Recovery: Benefits and Risks [Last Updated On: April 8th, 2025] [Originally Added On: April 8th, 2025]
- 0060) Androgel: A Promising Treatment for Psoriasis in American Men [Last Updated On: April 9th, 2025] [Originally Added On: April 9th, 2025]
- 0061) Androgel Use and Acne: Causes, Management, and Prevention Strategies for American Men [Last Updated On: April 9th, 2025] [Originally Added On: April 9th, 2025]
- 0062) Androgel's Role in Hair Regrowth for American Men with Alopecia: Benefits and Risks [Last Updated On: April 10th, 2025] [Originally Added On: April 10th, 2025]
- 0063) Androgel Use and Shingles Management in American Men: A Comprehensive Guide [Last Updated On: April 10th, 2025] [Originally Added On: April 10th, 2025]
- 0064) Androgel's Potential in Managing Herpes Outbreaks in American Men [Last Updated On: April 11th, 2025] [Originally Added On: April 11th, 2025]
- 0065) Androgel's Potential in Treating Vitiligo: Insights for American Men [Last Updated On: April 11th, 2025] [Originally Added On: April 11th, 2025]
- 0066) Androgel Enhances Recovery and Quality of Life in American Men with Burns [Last Updated On: April 12th, 2025] [Originally Added On: April 12th, 2025]
- 0067) Androgel's Role in Enhancing HIV Symptom Management in American Men [Last Updated On: April 15th, 2025] [Originally Added On: April 15th, 2025]
- 0068) Androgel's Potential Role in Enhancing Ebola Treatment for American Men [Last Updated On: April 16th, 2025] [Originally Added On: April 16th, 2025]
- 0069) Androgel's Role in Managing Zika Virus Symptoms in American Men: A Comprehensive Overview [Last Updated On: April 16th, 2025] [Originally Added On: April 16th, 2025]
- 0070) Androgel's Potential Role in TB Treatment for American Men: A Research Overview [Last Updated On: April 16th, 2025] [Originally Added On: April 16th, 2025]
- 0071) Androgel: A Potential Aid for American Men with Chikungunya Symptoms [Last Updated On: April 17th, 2025] [Originally Added On: April 17th, 2025]
- 0072) Androgel's Potential in Enhancing Immune Response Against Malaria in American Men [Last Updated On: April 18th, 2025] [Originally Added On: April 18th, 2025]
- 0073) Androgel Use and West Nile Virus: Impacts on Men's Health in the U.S. [Last Updated On: April 18th, 2025] [Originally Added On: April 18th, 2025]
- 0074) Androgel's Potential Role in Hepatitis C Treatment for American Men [Last Updated On: April 19th, 2025] [Originally Added On: April 19th, 2025]
- 0075) Androgel's Potential in Treating Yellow Fever: Benefits and Implications for American Men [Last Updated On: April 19th, 2025] [Originally Added On: April 19th, 2025]
- 0076) Androgel: A New Hope for American Men Battling Lyme Disease Symptoms [Last Updated On: April 19th, 2025] [Originally Added On: April 19th, 2025]
- 0077) Androgel's Role in Enhancing Dengue Recovery for American Men: A Comprehensive Overview [Last Updated On: April 19th, 2025] [Originally Added On: April 19th, 2025]
- 0078) Androgel's Potential in Managing Rabies Symptoms in American Men: A Novel Approach [Last Updated On: April 21st, 2025] [Originally Added On: April 21st, 2025]
- 0079) Androgel's Impact on Influenza Recovery in American Men: Benefits and Precautions [Last Updated On: April 21st, 2025] [Originally Added On: April 21st, 2025]
- 0080) Androgel: Enhancing American Men's Vitality and Well-being Through Testosterone Therapy [Last Updated On: April 22nd, 2025] [Originally Added On: April 22nd, 2025]
Word Count: 293