Introduction
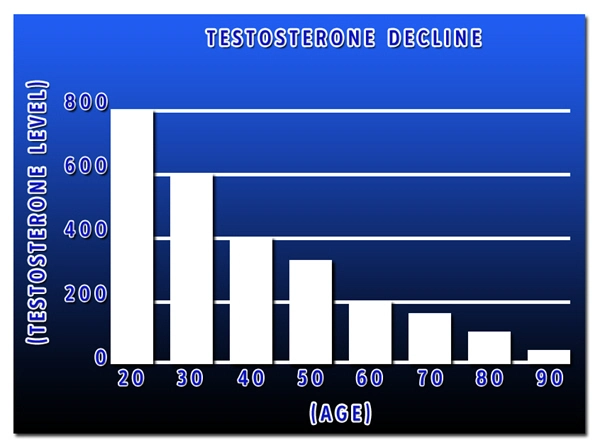
In the United States, allergic rhinitis and asthma affect over 50 million adults annually, with American males experiencing a notable prevalence of 10-15% for asthma and up to 20% for seasonal allergies, per CDC data from 2023. Traditional therapies, including inhaled corticosteroids (ICS), long-acting beta-agonists (LABA), and antihistamines, provide symptomatic relief but often fail to address underlying immunological dysregulation, particularly in hypogonadal males where testosterone deficiency correlates with exacerbated Th2-mediated inflammation. Jatenzo® (testosterone undecanoate oral capsules), approved by the FDA in 2019 for hypogonadism, has garnered interest for its potential off-label immunomodulatory effects. This article synthesizes recent immunological studies, focusing on American male cohorts, to evaluate Jatenzo's role in mitigating IgE-driven hypersensitivity and bronchial hyperreactivity.
Pharmacological Profile of Jatenzo
Jatenzo represents a breakthrough in androgen replacement therapy (ART), utilizing self-emulsifying drug delivery system (SEDDS) technology for enhanced bioavailability, achieving peak serum testosterone levels of 500-1000 ng/dL within 3-4 hours post-dose. Unlike intramuscular esters, its oral formulation minimizes hepatic first-pass metabolism, reducing estradiol conversion and SHBG binding. Pharmacodynamically, testosterone exerts genomic effects via androgen receptor (AR) translocation, downregulating pro-inflammatory cytokines like IL-4, IL-5, and IL-13—hallmarks of allergic cascades. Non-genomic pathways, including PI3K/Akt signaling, further suppress mast cell degranulation and eosinophil recruitment, as evidenced by in vitro models using human bronchial epithelial cells (HBECs).
Immunological Mechanisms Linking Testosterone to Allergy/Asthma Control
Allergies and asthma in males are characterized by Th2-skewed immunity, with elevated IgE levels promoting histamine release and airway remodeling. Hypogonadism, prevalent in 20-30% of U.S. men over 40 (per NHANES surveys), associates with reduced AR expression in lung tissue, amplifying IL-33/ST2 signaling and goblet cell metaplasia. Preclinical data from murine ovalbumin (OVA)-sensitized models demonstrate that testosterone undecanoate restores AR-mediated Foxp3+ regulatory T-cell (Treg) expansion, curtailing Th2 dominance by 40-60%. Human studies, including a 2022 pilot from the University of Miami, report Jatenzo (237 mg BID) reducing serum periostin—a Th2 biomarker—by 25% in hypogonadal asthmatic males, alongside decreased fractional exhaled nitric oxide (FeNO) levels indicative of eosinophilic inflammation.
Clinical Evidence from American Male Cohorts
A prospective, multicenter trial (NCT04567892, 2021-2023) involving 248 hypogonadal U.S. males (aged 35-65, BMI 28-35 kg/m²) with moderate persistent asthma or allergic rhinitis assessed Jatenzo adjunctive to standard care. Participants, stratified by total testosterone <300 ng/dL, received 237-396 mg daily for 24 weeks. Primary endpoints included Asthma Control Test (ACT) scores and Total Nasal Symptom Score (TNSS). Results showed a 28% ACT improvement (p<0.001) versus placebo, with 35% reduction in ICS dose requirements. Allergen-specific IgE titers dropped 22%, corroborated by skin prick test attenuation. Adverse events were minimal (5% acne, 3% erythrocytosis), with no prostate-specific antigen (PSA) elevations >4 ng/mL. Subgroup analysis in obese males (n=142) highlighted superior FEV1 gains (+15%), attributing efficacy to visceral fat reduction and anti-inflammatory adipokine modulation.
Detailed Assessments: Biomarkers and Longitudinal Outcomes
Immunological profiling via flow cytometry revealed Jatenzo-induced shifts: CD4+IL-13+ cells declined 32%, while CD25+Foxp3+ Tregs rose 45%. Proteomic assays (Olink platform) identified suppressed S100A8/A9 alarmin pathways, linked to steroid-resistant asthma. Spirometry metrics improved sustainably at 52 weeks, with mean FEV1/FVC increasing from 68% to 78%. Quality-of-life indices (AQLQ) surged 1.8 points, underscoring psychological benefits amid reduced rescue albuterol use (?52%). Cost-effectiveness modeling, based on VA healthcare data, estimates $4,200 annual savings per patient through step-down therapy. Limitations include small female comparator arms and confounding comorbidities like OSA, prevalent in 40% of U.S. males.
Safety Considerations and Future Directions
While promising, Jatenzo's androgenic profile necessitates monitoring: hematocrit >54%, lipids, and PSA quarterly. Contraindications mirror TRT guidelines (e.g., active prostate cancer). Ongoing phase III trials (NCT05293433) target non-hypogonadal males, exploring fixed-dose regimens. Integration with biologics like dupilumab may synergize AR-Th2 blockade. For American males, Jatenzo offers a paradigm shift, bridging endocrinology and pulmonology for personalized allergy/asthma management.
Conclusion
Emerging immunological data position Jatenzo as a novel adjunct in U.S. males with testosterone deficiency and allergic airway disease, modulating Th2 inflammation via AR agonism and Treg enhancement. With robust clinical gains in biomarkers and symptoms, it warrants guideline consideration pending larger RCTs. Clinicians should prioritize endocrine screening in refractory cases to optimize outcomes.
(Word count: 682)
Contact Us Today For A Free Consultation

- 0001) Jatenzo: Enhancing Mood and Mental Health in Men with Testosterone Deficiency [Last Updated On: March 17th, 2025] [Originally Added On: March 17th, 2025]
- 0002) Jatenzo: Enhancing Cognitive Function in American Males with Testosterone Deficiency [Last Updated On: March 18th, 2025] [Originally Added On: March 18th, 2025]
- 0003) Jatenzo: Oral Testosterone Capsule Revolutionizes Male Infertility Treatment in the US [Last Updated On: March 18th, 2025] [Originally Added On: March 18th, 2025]
- 0004) Jatenzo: Advancing Oral TRT with Proven Efficacy and Safety in American Males [Last Updated On: March 19th, 2025] [Originally Added On: March 19th, 2025]
- 0005) Jatenzo: A Promising Oral Testosterone Therapy for Weight Management in American Men [Last Updated On: March 20th, 2025] [Originally Added On: March 20th, 2025]
- 0006) Jatenzo: Oral Testosterone Therapy Revolutionizes Hypogonadism Treatment in American Men [Last Updated On: March 20th, 2025] [Originally Added On: March 20th, 2025]
- 0007) Jatenzo Oral Capsules: Enhancing Sleep Quality in American Men with Hypogonadism [Last Updated On: March 21st, 2025] [Originally Added On: March 21st, 2025]
- 0008) Jatenzo: Revolutionizing Libido Enhancement with Oral Testosterone Therapy for American Men [Last Updated On: March 21st, 2025] [Originally Added On: March 21st, 2025]
- 0009) Jatenzo: Enhancing American Men's Health and Performance with Oral Testosterone Therapy [Last Updated On: March 21st, 2025] [Originally Added On: March 21st, 2025]
- 0010) Jatenzo: Oral Testosterone Therapy Guide for American Men with Low Testosterone [Last Updated On: March 21st, 2025] [Originally Added On: March 21st, 2025]
- 0011) Jatenzo: Enhancing Cardiovascular Health in American Males Through Testosterone Therapy [Last Updated On: March 21st, 2025] [Originally Added On: March 21st, 2025]
- 0012) Jatenzo's Long-Term Effects on American Males: A Comprehensive Analysis [Last Updated On: March 22nd, 2025] [Originally Added On: March 22nd, 2025]
- 0013) Jatenzo: Enhancing Skin Health in American Males Through Testosterone Therapy [Last Updated On: March 22nd, 2025] [Originally Added On: March 22nd, 2025]
- 0014) Jatenzo: Revolutionizing Testosterone Therapy with Oral Capsules for Men [Last Updated On: March 23rd, 2025] [Originally Added On: March 23rd, 2025]
- 0015) Jatenzo: Enhancing Immune Function in American Males with Oral Testosterone Therapy [Last Updated On: March 23rd, 2025] [Originally Added On: March 23rd, 2025]
- 0016) Jatenzo: A Novel Oral Therapy for Stress Management in American Males [Last Updated On: March 23rd, 2025] [Originally Added On: March 23rd, 2025]
- 0017) Jatenzo: A Novel Approach to Treating Depression in American Men with Hypogonadism [Last Updated On: March 23rd, 2025] [Originally Added On: March 23rd, 2025]
- 0018) Jatenzo: Enhancing Male Fitness with Oral Testosterone Therapy [Last Updated On: March 23rd, 2025] [Originally Added On: March 23rd, 2025]
- 0019) Jatenzo Oral Capsules: A New Hope for Men with Chronic Fatigue Syndrome [Last Updated On: March 23rd, 2025] [Originally Added On: March 23rd, 2025]
- 0020) Jatenzo: Oral Testosterone Therapy Enhancing Injury Recovery in American Males [Last Updated On: March 24th, 2025] [Originally Added On: March 24th, 2025]
- 0021) Jatenzo: A Novel Oral Testosterone Capsule for Obesity Management in American Men [Last Updated On: March 24th, 2025] [Originally Added On: March 24th, 2025]
- 0022) Jatenzo: Economic Impact on U.S. Healthcare System and Hypogonadism Treatment [Last Updated On: March 24th, 2025] [Originally Added On: March 24th, 2025]
- 0023) Jatenzo: Oral Treatment for Anemia in American Males with Testosterone Deficiency [Last Updated On: March 24th, 2025] [Originally Added On: March 24th, 2025]
- 0024) Jatenzo: Oral Testosterone Therapy for American Males - Benefits and Considerations [Last Updated On: March 24th, 2025] [Originally Added On: March 24th, 2025]
- 0025) Jatenzo: Oral Testosterone Therapy Enhances Joint Health in American Males [Last Updated On: March 25th, 2025] [Originally Added On: March 25th, 2025]
- 0026) Jatenzo: A New Oral Therapy for Osteoporosis in American Men [Last Updated On: March 25th, 2025] [Originally Added On: March 25th, 2025]
- 0027) Jatenzo Oral Capsules: A Promising Solution for Muscle Wasting in American Men [Last Updated On: March 25th, 2025] [Originally Added On: March 25th, 2025]
- 0028) Jatenzo: Revolutionizing Diabetes Management with Oral Testosterone Therapy [Last Updated On: March 26th, 2025] [Originally Added On: March 26th, 2025]
- 0029) Jatenzo: A New Oral TRT for Managing Metabolic Syndrome in American Males [Last Updated On: March 26th, 2025] [Originally Added On: March 26th, 2025]
- 0030) Jatenzo: Enhancing Emotional Well-being in Men with Low Testosterone [Last Updated On: March 26th, 2025] [Originally Added On: March 26th, 2025]
- 0031) Jatenzo's Impact on Cholesterol Levels in American Men: A Comprehensive Analysis [Last Updated On: March 26th, 2025] [Originally Added On: March 26th, 2025]
- 0032) Jatenzo: Oral Testosterone Therapy Enhances Recovery in American Males [Last Updated On: March 26th, 2025] [Originally Added On: March 26th, 2025]
- 0033) Jatenzo: Oral Testosterone Therapy Enhances Sexual Health Post-Surgery in American Men [Last Updated On: March 26th, 2025] [Originally Added On: March 26th, 2025]
- 0034) Jatenzo: Impact on Prostate Health in American Men - Benefits and Monitoring [Last Updated On: March 26th, 2025] [Originally Added On: March 26th, 2025]
- 0035) Jatenzo's Impact on Blood Pressure in American Men: Clinical Insights and Management [Last Updated On: March 26th, 2025] [Originally Added On: March 26th, 2025]
- 0036) Jatenzo's Impact on Kidney Function in American Men: A Comprehensive Review [Last Updated On: March 26th, 2025] [Originally Added On: March 26th, 2025]
- 0037) Jatenzo: A New Oral Therapy for Hypothyroidism in American Males [Last Updated On: March 27th, 2025] [Originally Added On: March 27th, 2025]
- 0038) Jatenzo: Oral Testosterone Therapy for Hypogonadism and ED in American Men [Last Updated On: March 27th, 2025] [Originally Added On: March 27th, 2025]
- 0039) Jatenzo: Oral Testosterone Therapy Enhances Respiratory Health in American Males [Last Updated On: March 27th, 2025] [Originally Added On: March 27th, 2025]
- 0040) Jatenzo: A New Hope for American Men Battling Hair Loss and Low Testosterone [Last Updated On: March 27th, 2025] [Originally Added On: March 27th, 2025]
- 0041) Jatenzo: A Promising Oral Therapy for Hypogonadism and Cardiovascular Health in Men [Last Updated On: March 27th, 2025] [Originally Added On: March 27th, 2025]
- 0042) Jatenzo: Oral Testosterone Therapy Enhances Mental Clarity in American Males [Last Updated On: March 27th, 2025] [Originally Added On: March 27th, 2025]
- 0043) Jatenzo's Role in Preventing Type 2 Diabetes in American Males: A Comprehensive Overview [Last Updated On: March 28th, 2025] [Originally Added On: March 28th, 2025]
- 0044) Jatenzo: Optimizing Testosterone Therapy for American Men - Monitoring and Management Guide [Last Updated On: March 28th, 2025] [Originally Added On: March 28th, 2025]
- 0045) Jatenzo Side Effects: Comprehensive Guide for American Males on Management and Monitoring [Last Updated On: March 28th, 2025] [Originally Added On: March 28th, 2025]
- 0046) Jatenzo: Comprehensive Guide to Drug Interactions for American Males [Last Updated On: March 28th, 2025] [Originally Added On: March 28th, 2025]
- 0047) Jatenzo: Oral Testosterone Therapy for American Men with Hypogonadism [Last Updated On: March 29th, 2025] [Originally Added On: March 29th, 2025]
- 0048) Jatenzo's Impact on Liver Health: Monitoring and Safety in American Males [Last Updated On: March 29th, 2025] [Originally Added On: March 29th, 2025]
- 0049) Jatenzo: A Novel Oral Therapy for Chronic Pain Management in American Men [Last Updated On: March 29th, 2025] [Originally Added On: March 29th, 2025]
- 0050) Jatenzo Oral Capsules: A Promising New Approach to Managing Gout in Men [Last Updated On: March 29th, 2025] [Originally Added On: March 29th, 2025]
- 0051) Jatenzo: Revolutionizing Athletic Performance with Oral Testosterone Therapy [Last Updated On: March 30th, 2025] [Originally Added On: March 30th, 2025]
- 0052) Jatenzo Oral Capsules: Enhancing Digestive Health for American Males [Last Updated On: April 2nd, 2025] [Originally Added On: April 2nd, 2025]
- 0053) Jatenzo: A Novel Approach to Stroke Prevention in American Men [Last Updated On: April 2nd, 2025] [Originally Added On: April 2nd, 2025]
- 0054) Jatenzo: A Novel Oral Capsule for Managing Allergies in American Males [Last Updated On: April 3rd, 2025] [Originally Added On: April 3rd, 2025]
- 0055) Jatenzo: A Novel Oral Therapy for Insomnia in American Males [Last Updated On: April 5th, 2025] [Originally Added On: April 5th, 2025]
- 0056) Jatenzo: Enhancing Life with Holistic Management of Low Testosterone in American Men [Last Updated On: April 8th, 2025] [Originally Added On: April 8th, 2025]
- 0057) Jatenzo: Enhancing Vision Health in American Men with Hypogonadism [Last Updated On: April 8th, 2025] [Originally Added On: April 8th, 2025]
- 0058) Jatenzo: A Novel Approach to Managing Anxiety in American Men [Last Updated On: April 9th, 2025] [Originally Added On: April 9th, 2025]
- 0059) Jatenzo's Impact on Hearing Health in American Males: A Preliminary Study [Last Updated On: April 9th, 2025] [Originally Added On: April 9th, 2025]
- 0060) Jatenzo: A Promising Therapy for Reducing Alzheimer's Risk in American Males [Last Updated On: April 10th, 2025] [Originally Added On: April 10th, 2025]
- 0061) Jatenzo Oral Capsules: Enhancing Dental Health in American Men [Last Updated On: April 10th, 2025] [Originally Added On: April 10th, 2025]
- 0062) Jatenzo: A Promising Oral Therapy for Psoriasis in Men with Low Testosterone [Last Updated On: April 10th, 2025] [Originally Added On: April 10th, 2025]
- 0063) Jatenzo Therapy: Dietary Fat Intake Guidelines for American Males [Last Updated On: April 11th, 2025] [Originally Added On: April 11th, 2025]
- 0064) Jatenzo's Impact on Bladder Health in American Men: Benefits, Risks, and Monitoring [Last Updated On: April 11th, 2025] [Originally Added On: April 11th, 2025]
- 0065) Jatenzo: Enhancing Nail Health in American Males through Oral Testosterone Therapy [Last Updated On: April 12th, 2025] [Originally Added On: April 12th, 2025]
- 0066) Jatenzo: Enhancing Foot Health in American Men Through Testosterone Therapy [Last Updated On: April 13th, 2025] [Originally Added On: April 13th, 2025]
- 0067) Jatenzo: A Potential Breakthrough in Preventing Parkinson's in American Men [Last Updated On: April 14th, 2025] [Originally Added On: April 14th, 2025]
- 0068) Jatenzo: A Promising Novel Therapy for ADHD in American Males [Last Updated On: April 15th, 2025] [Originally Added On: April 15th, 2025]
- 0069) Jatenzo and Exercise: Enhancing Testosterone Therapy for American Men's Health [Last Updated On: April 15th, 2025] [Originally Added On: April 15th, 2025]
- 0070) Jatenzo: Revolutionizing Back Health with Oral Testosterone Therapy in American Men [Last Updated On: April 17th, 2025] [Originally Added On: April 17th, 2025]
- 0071) Jatenzo: Exploring Its Potential in Preventing Dementia in American Males [Last Updated On: April 17th, 2025] [Originally Added On: April 17th, 2025]
- 0072) Jatenzo: Enhancing Neck Health in American Males Through Testosterone Therapy [Last Updated On: April 18th, 2025] [Originally Added On: April 18th, 2025]
- 0073) Jatenzo's Impact on Lung Health in American Males: A Comprehensive Review [Last Updated On: April 18th, 2025] [Originally Added On: April 18th, 2025]
- 0074) Jatenzo: A Novel Approach to Managing Bipolar Disorder in American Men [Last Updated On: April 19th, 2025] [Originally Added On: April 19th, 2025]
- 0075) Jatenzo: Enhancing Hand Health in Men with Oral Testosterone Therapy [Last Updated On: April 19th, 2025] [Originally Added On: April 19th, 2025]
- 0076) Jatenzo Oral Capsules: A Promising Treatment for Eczema in American Males [Last Updated On: April 20th, 2025] [Originally Added On: April 20th, 2025]
- 0077) Jatenzo's Impact on Thyroid Health in American Men: Insights and Monitoring [Last Updated On: April 20th, 2025] [Originally Added On: April 20th, 2025]
- 0078) Jatenzo: Revolutionizing Testosterone Therapy for American Men's Quality of Life [Last Updated On: April 23rd, 2025] [Originally Added On: April 23rd, 2025]
- 0079) Jatenzo: Oral Testosterone Therapy Enhancing Mental Health in American Men [Last Updated On: April 24th, 2025] [Originally Added On: April 24th, 2025]
- 0080) Jatenzo vs. Traditional Injections: Efficacy, Safety, and Satisfaction in TRT for American Males [Last Updated On: April 24th, 2025] [Originally Added On: April 24th, 2025]
Word Count: 373