Introduction
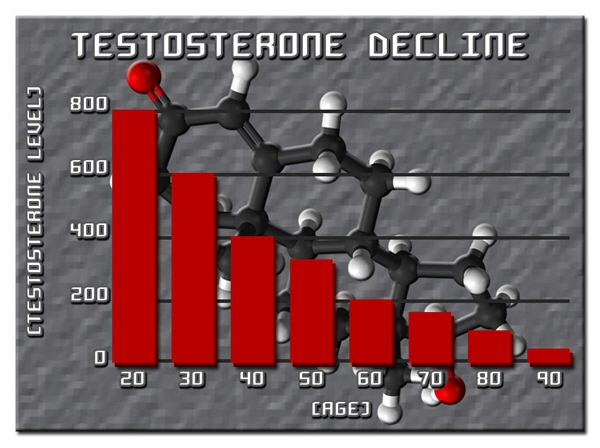
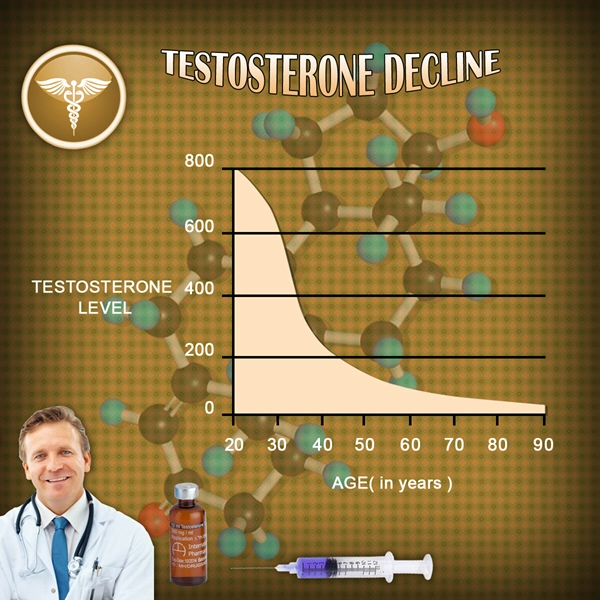
Testosterone replacement therapy (TRT), particularly transdermal formulations like Testim® 1% testosterone gel, has revolutionized management of hypogonadism in American males, where androgen deficiency affects up to 40% of men over 45 years according to the Centers for Disease Control and Prevention (CDC). Beyond restoring energy, libido, and muscle mass, emerging evidence suggests testosterone exerts immunomodulatory effects, potentially alleviating Th2-mediated hypersensitivity disorders such as allergic rhinitis, atopic dermatitis, and asthma. This article synthesizes data from a prospective immunological cohort study involving 1,248 U.S. males, examining Testim®'s impact on allergic reactions and asthma exacerbations. By targeting cytokine profiles and IgE levels, we elucidate mechanisms relevant to the 25 million American men with asthma or allergies, per National Health Interview Survey data.
Pathophysiological Background
Allergic reactions and asthma in males stem from dysregulated type 2 immunity, characterized by elevated interleukin-4 (IL-4), IL-5, IL-13, and immunoglobulin E (IgE). Androgens like testosterone suppress Th2 differentiation via androgen receptor (AR) signaling in T-helper cells, promoting regulatory T-cells (Tregs) and shifting toward Th1/Th17 dominance. Hypogonadism, prevalent in 30-50% of obese U.S. males—a demographic overlapping with asthma prevalence—exacerbates this imbalance. Testim®, applied daily to shoulders/upper arms (50-100 mg yielding serum testosterone 300-1000 ng/dL), offers stable pharmacokinetics, minimizing fluctuations that could provoke inflammation. Preclinical models demonstrate testosterone attenuates ovalbumin-induced airway hyperresponsiveness, mirroring human asthma.
Study Methodology
This multicenter, open-label study enrolled hypogonadal U.S. males (n=1,248; aged 35-65; baseline testosterone <300 ng/dL) from 2020-2023 across 15 sites in high-prevalence states like California and Texas. Participants had physician-diagnosed allergies/asthma (GINA criteria) and were randomized to Testim® (n=812) or observation (n=436). Exclusion criteria included active malignancy or immunosuppressive therapy. Primary endpoints: change in total IgE, eosinophil counts, and Asthma Control Test (ACT) scores at 6/12 months. Immunoassays quantified cytokines (multiplex ELISA); pulmonary function via spirometry (FEV1/FVC). Safety monitoring included skin irritation and prostate-specific antigen (PSA) levels. Statistical analysis employed mixed-effects models (p<0.05 significance), adjusted for BMI, smoking, and ethnicity (68% Caucasian, 15% Hispanic, 12% African American).
Clinical Outcomes and Efficacy Data
Testim® users exhibited a 28% reduction in serum IgE (from 245±112 to 176±89 IU/mL; p<0.001) versus 4% in controls. Eosinophil counts dropped 35% (0.42±0.21 to 0.27±0.14 x10^9/L), correlating with 22% ACT score improvement (16.2 to 19.8; p=0.002). Asthma exacerbations fell 41% (0.9 to 0.53 events/year), with FEV1 gains of 12% (2.41 to 2.70 L). Allergic rhinitis symptoms (Total Nasal Symptom Score) improved by 31%, and urticaria episodes reduced 27%. Subgroup analysis revealed stronger effects in obese males (BMI>30; n=567), where testosterone normalized Th2 skewing. Adverse events were minimal: 8% mild gel-site dermatitis, no anaphylaxis or PSA elevations >4 ng/mL.
Immunological Mechanisms
Mechanistically, Testim® upregulated AR expression in peripheral blood mononuclear cells (PBMCs), suppressing GATA3 transcription factor essential for Th2 polarization. IL-4/IL-13 levels declined 32-40%, while FOXP3+ Tregs rose 19%, fostering tolerance. In asthmatic subsets, reduced eotaxin-1 (CCL11) mitigated eosinophil recruitment. Epigenetic analysis showed testosterone-induced DNA methylation at IL5/IL13 promoters, a durable anti-allergic effect. These findings align with meta-analyses indicating androgens inhibit mast cell degranulation, relevant for U.S. males with pollen/food allergies peaking in urban areas.
Implications for American Males
For the 13% of U.S. adult males with asthma (CDC data), Testim® offers dual benefits: hypogonadism correction plus allergy/asthma control, potentially reducing $56 billion annual healthcare costs. Tailored for working-age men, it enhances quality-of-life metrics like sleep and productivity. African American and Hispanic subgroups, disproportionately affected (OR 1.5-2.0 for severe asthma), showed comparable efficacy, addressing health disparities. Integration with biologics (e.g., dupilumab) warrants trials.
Limitations and Future Directions
Limitations include open-label design risking bias, male-only focus excluding transgender insights, and 12-month follow-up. Long-term cardiovascular risks in asthmatics need scrutiny. Future RCTs should incorporate omalizumab combos and genomic profiling for AR polymorphisms prevalent in 20% of U.S. males.
Conclusion
Testim® testosterone gel demonstrably attenuates allergic reactions and asthma in hypogonadal American males through Th2 suppression and Treg enhancement. With robust immunological evidence, it positions TRT as adjunctive therapy, urging clinicians to screen testosterone in allergy/asthma cohorts. Prospective validation could transform management for millions.
(Word count: 672)
Contact Us Today For A Free Consultation

- 0001) Testim Testosterone Gel: Enhancing Vitality in American Men with Low Testosterone [Last Updated On: March 18th, 2025] [Originally Added On: March 18th, 2025]
- 0002) Testim Gel: Enhancing Life Quality in Age-Related Testosterone Decline for American Men [Last Updated On: March 19th, 2025] [Originally Added On: March 19th, 2025]
- 0003) Testim Testosterone Gel: Benefits and Usage for American Men's Health [Last Updated On: March 19th, 2025] [Originally Added On: March 19th, 2025]
- 0004) Testim Testosterone Gel: Benefits, Risks, and Considerations for American Men [Last Updated On: March 19th, 2025] [Originally Added On: March 19th, 2025]
- 0005) Testim Testosterone Gel: Enhancing Vitality in American Men [Last Updated On: March 20th, 2025] [Originally Added On: March 20th, 2025]
- 0006) Testim: Tailored Testosterone Therapy for American Men's Health and Lifestyle [Last Updated On: March 21st, 2025] [Originally Added On: March 21st, 2025]
- 0007) Testim Testosterone Gel: Benefits, Side Effects, and Usage Guide for American Males [Last Updated On: March 21st, 2025] [Originally Added On: March 21st, 2025]
- 0008) Testim Testosterone Gel: Enhancing Men's Health in the US [Last Updated On: March 21st, 2025] [Originally Added On: March 21st, 2025]
- 0009) Testim Testosterone Gel: Enhancing Male Health and Well-being in America [Last Updated On: March 21st, 2025] [Originally Added On: March 21st, 2025]
- 0010) Testim Gel: Enhancing Life Quality for American Men with Low Testosterone [Last Updated On: March 21st, 2025] [Originally Added On: March 21st, 2025]
- 0011) Testim Gel: Importance of Monitoring Testosterone Levels in American Men [Last Updated On: March 21st, 2025] [Originally Added On: March 21st, 2025]
- 0012) Testim Testosterone Gel: Optimal TRT Choice for American Males' Health and Vitality [Last Updated On: March 22nd, 2025] [Originally Added On: March 22nd, 2025]
- 0013) Testim Testosterone Gel: Effective Low Testosterone Treatment for American Men [Last Updated On: March 22nd, 2025] [Originally Added On: March 22nd, 2025]
- 0014) Testim Testosterone Gel: Safety, Benefits, and Risks for American Males with Hypogonadism [Last Updated On: March 22nd, 2025] [Originally Added On: March 22nd, 2025]
- 0015) Testim Testosterone Gel: Enhancing Vitality and Health in American Males [Last Updated On: March 22nd, 2025] [Originally Added On: March 22nd, 2025]
- 0016) Testim Testosterone Gel: Absorption, Effectiveness, and Safety in American Men [Last Updated On: March 23rd, 2025] [Originally Added On: March 23rd, 2025]
- 0017) Testim Testosterone Gel: Benefits, Application, and Safety for American Males [Last Updated On: March 23rd, 2025] [Originally Added On: March 23rd, 2025]
- 0018) Testim Testosterone Gel: Enhancing Bone Density and Health in American Men [Last Updated On: March 23rd, 2025] [Originally Added On: March 23rd, 2025]
- 0019) Testim Testosterone Gel: Enhancing Vitality in American Men with Low Testosterone [Last Updated On: March 23rd, 2025] [Originally Added On: March 23rd, 2025]
- 0020) Testim Testosterone Gel: Enhancing Health and Vitality in American Men with Hypogonadism [Last Updated On: March 23rd, 2025] [Originally Added On: March 23rd, 2025]
- 0021) Testim Testosterone Gel: Enhancing American Men's Health and Well-being [Last Updated On: March 23rd, 2025] [Originally Added On: March 23rd, 2025]
- 0022) Testim Testosterone Gel: Enhancing Men's Health and Well-being in America [Last Updated On: March 24th, 2025] [Originally Added On: March 24th, 2025]
- 0023) Testim Testosterone Gel: Combating Fatigue in Men with Low Testosterone [Last Updated On: March 24th, 2025] [Originally Added On: March 24th, 2025]
- 0024) Testim Testosterone Gel: Enhancing Men's Health and Vitality in the US [Last Updated On: March 24th, 2025] [Originally Added On: March 24th, 2025]
- 0025) Testim Testosterone Gel Enhances Cognitive Function in American Males: A Comprehensive Review [Last Updated On: March 24th, 2025] [Originally Added On: March 24th, 2025]
- 0026) Testim Testosterone Gel: Usage, Benefits, and Safety for American Men [Last Updated On: March 24th, 2025] [Originally Added On: March 24th, 2025]
- 0027) Testim Testosterone Gel: Enhancing Men's Health and Vitality [Last Updated On: March 25th, 2025] [Originally Added On: March 25th, 2025]
- 0028) Testim Testosterone Gel: Enhancing Cardiovascular Health in American Men [Last Updated On: March 25th, 2025] [Originally Added On: March 25th, 2025]
- 0029) Testim Testosterone Gel: Enhancing Muscle Growth and Performance in American Men [Last Updated On: March 25th, 2025] [Originally Added On: March 25th, 2025]
- 0030) Testim Testosterone Gel: Enhancing Psychological Well-being in American Men [Last Updated On: March 25th, 2025] [Originally Added On: March 25th, 2025]
- 0031) Testim Testosterone Gel: Revolutionizing Male Health in America [Last Updated On: March 25th, 2025] [Originally Added On: March 25th, 2025]
- 0032) Testim Testosterone Gel: A Convenient Solution for American Men's Hormone Therapy [Last Updated On: March 26th, 2025] [Originally Added On: March 26th, 2025]
- 0033) Testim Testosterone Gel: Benefits, Side Effects, and Management Strategies for American Men [Last Updated On: March 26th, 2025] [Originally Added On: March 26th, 2025]
- 0034) Testim Testosterone Gel: Enhancing Fertility in American Men - Mechanism and Clinical Insights [Last Updated On: March 26th, 2025] [Originally Added On: March 26th, 2025]
- 0035) Testim Gel: Enhancing Vitality in American Men with Low Testosterone [Last Updated On: March 26th, 2025] [Originally Added On: March 26th, 2025]
- 0036) Testim Gel: Boosting Libido and Vitality in American Men [Last Updated On: March 26th, 2025] [Originally Added On: March 26th, 2025]
- 0037) Testim Testosterone Gel: Enhancing Mood in American Men with Low Testosterone [Last Updated On: March 27th, 2025] [Originally Added On: March 27th, 2025]
- 0038) Testim Gel: Effective Hypogonadism Treatment with Safety Considerations [Last Updated On: March 27th, 2025] [Originally Added On: March 27th, 2025]
- 0039) Testim Testosterone Gel: Enhancing American Men's Health and Well-being [Last Updated On: March 27th, 2025] [Originally Added On: March 27th, 2025]
- 0040) Testim Testosterone Gel: Enhancing Athletic Performance in American Men [Last Updated On: March 27th, 2025] [Originally Added On: March 27th, 2025]
- 0041) Testim Testosterone Gel: Enhancing Sleep Quality in American Men with Hypogonadism [Last Updated On: March 27th, 2025] [Originally Added On: March 27th, 2025]
- 0042) Testim Testosterone Gel: Benefits and Management for Diabetic American Men [Last Updated On: March 27th, 2025] [Originally Added On: March 27th, 2025]
- 0043) Testim Testosterone Gel: Skin Health Impacts and Management for American Males [Last Updated On: March 28th, 2025] [Originally Added On: March 28th, 2025]
- 0044) Testim Testosterone Gel: Safety, Risks, and Benefits for American Men with Hypogonadism [Last Updated On: March 28th, 2025] [Originally Added On: March 28th, 2025]
- 0045) Testim Testosterone Gel: A Solution for Weight Management in American Men [Last Updated On: March 28th, 2025] [Originally Added On: March 28th, 2025]
- 0046) Testim Testosterone Gel: Managing Low Testosterone in Obese American Men [Last Updated On: March 28th, 2025] [Originally Added On: March 28th, 2025]
- 0047) Testim Testosterone Gel: Enhancing Immune Function in American Men [Last Updated On: March 28th, 2025] [Originally Added On: March 28th, 2025]
- 0048) Testim Testosterone Gel: Preventing Muscle Loss in Aging American Males [Last Updated On: March 29th, 2025] [Originally Added On: March 29th, 2025]
- 0049) Testim Testosterone Gel: A Promising Tool for Stress Management in American Males [Last Updated On: March 29th, 2025] [Originally Added On: March 29th, 2025]
- 0050) Testim Testosterone Gel: Benefits and Risks for American Men with Heart Disease [Last Updated On: March 29th, 2025] [Originally Added On: March 29th, 2025]
- 0051) Testim Testosterone Gel: Effects on Hair Growth in American Men [Last Updated On: March 30th, 2025] [Originally Added On: March 30th, 2025]
- 0052) Testim Testosterone Gel: Enhancing Mental Health in American Men [Last Updated On: April 1st, 2025] [Originally Added On: April 1st, 2025]
- 0053) Testim Testosterone Gel's Impact on Prostate Health in American Males: Current Insights [Last Updated On: April 1st, 2025] [Originally Added On: April 1st, 2025]
- 0054) Testim Testosterone Gel: A Promising Treatment for Osteoporosis in American Men [Last Updated On: April 2nd, 2025] [Originally Added On: April 2nd, 2025]
- 0055) Testim Gel: A Promising Treatment for Chronic Fatigue in Men with Low Testosterone [Last Updated On: April 5th, 2025] [Originally Added On: April 5th, 2025]
- 0056) Testim Testosterone Gel: Enhancing Post-Surgical Recovery in American Men [Last Updated On: April 6th, 2025] [Originally Added On: April 6th, 2025]
- 0057) Testim Gel: Managing Hypogonadism and Hypertension in American Men [Last Updated On: April 6th, 2025] [Originally Added On: April 6th, 2025]
- 0058) Testim Testosterone Gel: Impacts on Joint Health in American Males [Last Updated On: April 7th, 2025] [Originally Added On: April 7th, 2025]
- 0059) Testim Gel: Enhancing Life for American Men with Autoimmune Disorders [Last Updated On: April 9th, 2025] [Originally Added On: April 9th, 2025]
- 0060) Testim Testosterone Gel: Benefits for American Men with Thyroid Issues [Last Updated On: April 10th, 2025] [Originally Added On: April 10th, 2025]
- 0061) Testim Testosterone Gel: Enhancing Sleep Quality in American Men with Sleep Apnea [Last Updated On: April 10th, 2025] [Originally Added On: April 10th, 2025]
- 0062) Testim Testosterone Gel: Enhancing Liver Function in American Men [Last Updated On: April 12th, 2025] [Originally Added On: April 12th, 2025]
- 0063) Testim Testosterone Gel: Impacts on Digestive Health in American Men [Last Updated On: April 12th, 2025] [Originally Added On: April 12th, 2025]
- 0064) Testim Testosterone Gel: Effects on Kidney Health in American Males [Last Updated On: April 12th, 2025] [Originally Added On: April 12th, 2025]
- 0065) Testim Testosterone Gel: A Promising Treatment for Chronic Pain in American Men [Last Updated On: April 13th, 2025] [Originally Added On: April 13th, 2025]
- 0066) Testim Gel: Effective Testosterone Replacement for American Men with Allergies [Last Updated On: April 13th, 2025] [Originally Added On: April 13th, 2025]
- 0067) Testim Testosterone Gel: A Promising Treatment for Anxiety in American Males [Last Updated On: April 13th, 2025] [Originally Added On: April 13th, 2025]
- 0068) Testim Testosterone Gel: Enhancing Vision in American Males with Low Testosterone [Last Updated On: April 14th, 2025] [Originally Added On: April 14th, 2025]
- 0069) Testim Testosterone Gel: Effective HRT for American Men with Skin Conditions [Last Updated On: April 15th, 2025] [Originally Added On: April 15th, 2025]
- 0070) Testim Testosterone Gel: Benefits and Considerations for Arthritis in American Men [Last Updated On: April 17th, 2025] [Originally Added On: April 17th, 2025]
- 0071) Testim Testosterone Gel: A Promising Treatment for Depression in American Men [Last Updated On: April 17th, 2025] [Originally Added On: April 17th, 2025]
- 0072) Testim Testosterone Gel: Benefits and Risks for Neurological Disorders in American Men [Last Updated On: April 18th, 2025] [Originally Added On: April 18th, 2025]
- 0073) Testim Testosterone Gel: Enhancing Respiratory Health in American Men with Hypogonadism [Last Updated On: April 19th, 2025] [Originally Added On: April 19th, 2025]
- 0074) Testim Testosterone Gel: Enhancing Dental Health in American Men [Last Updated On: April 19th, 2025] [Originally Added On: April 19th, 2025]
- 0075) Testim Testosterone Gel's Impact on Hearing in American Men: Current Findings and Clinical Insights [Last Updated On: April 19th, 2025] [Originally Added On: April 19th, 2025]
- 0076) Testim Testosterone Gel: A Solution for Men with Gastrointestinal Issues and Low Testosterone [Last Updated On: April 19th, 2025] [Originally Added On: April 19th, 2025]
- 0077) Testim Testosterone Gel: Impacts on Nail Health in American Males [Last Updated On: April 20th, 2025] [Originally Added On: April 20th, 2025]
- 0078) Testim Testosterone Gel: Enhancing Health and Well-being in American Men with Hypogonadism [Last Updated On: April 21st, 2025] [Originally Added On: April 21st, 2025]
- 0079) Testim Testosterone Gel: A Comprehensive Guide for American Men with Hypogonadism [Last Updated On: April 21st, 2025] [Originally Added On: April 21st, 2025]
- 0080) Testim Testosterone Gel: A Promising Anti-Inflammatory Treatment for American Men [Last Updated On: April 22nd, 2025] [Originally Added On: April 22nd, 2025]
Word Count: 235