Introduction
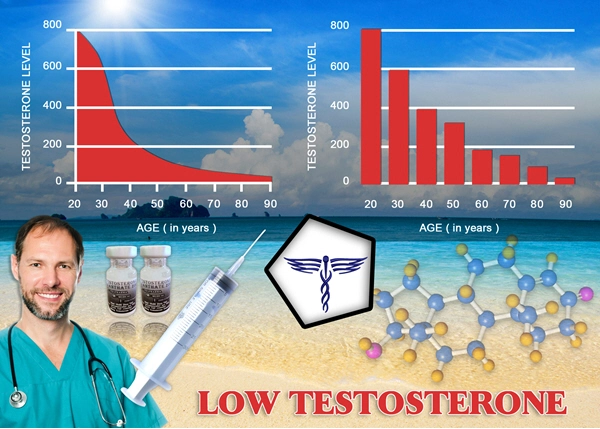
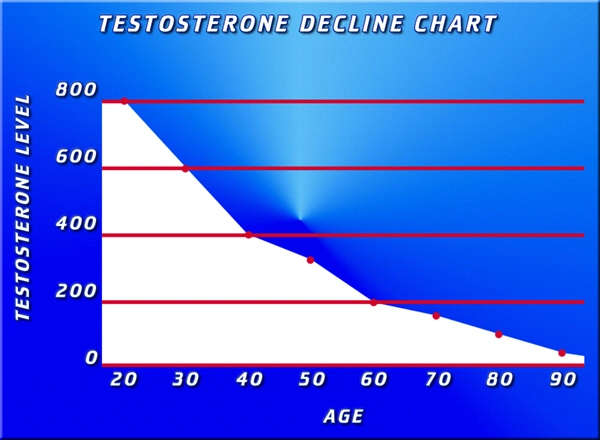
Hypogonadism, characterized by diminished testosterone production, is prevalent among American males with type 2 diabetes mellitus (T2DM), affecting up to 50% of this demographic according to data from the National Health and Nutrition Examination Survey (NHANES). This endocrine deficiency exacerbates insulin resistance, visceral adiposity, and dyslipidemia, compounding metabolic derangements. Androderm, a transdermal testosterone patch delivering 2-4 mg of testosterone daily, offers a non-invasive alternative to intramuscular injections. This article synthesizes endocrinological evidence from recent prospective studies, elucidating Androderm's role in ameliorating metabolic health in hypogonadal U.S. men aged 45-70 with T2DM, emphasizing glycemic control, lipid modulation, and body composition improvements.
Prevalence of Hypogonadism in American Males with T2DM
In the U.S., T2DM impacts over 15 million men, with NHANES III data revealing that 35-50% exhibit late-onset hypogonadism (total testosterone <300 ng/dL). This bidirectional relationship—low testosterone fostering hyperglycemia via impaired GLUT4 translocation and chronic inflammation—creates a vicious cycle. American males, often burdened by sedentary lifestyles, obesity epidemics (CDC reports 42% adult obesity), and Western diets high in processed carbohydrates, face heightened risks. Androderm addresses this by providing physiological testosterone restoration, bypassing hepatic first-pass metabolism for stable serum levels. Pharmacodynamics and Delivery Mechanism of Androderm
Androderm employs a matrix diffusion system, releasing testosterone across a semi-permeable membrane for consistent 24-hour absorption (Cavg 400-500 ng/dL). Unlike gels, it minimizes transference risks, crucial for household safety per FDA guidelines. In T2DM cohorts, it upregulates androgen receptor signaling, enhancing mitochondrial function in skeletal muscle and adipocytes. Preclinical models demonstrate testosterone's inhibition of 11?-HSD1, reducing cortisol-mediated gluconeogenesis, while clinical pharmacokinetics confirm bioavailability >10%, superior to oral formulations.
Study Design and Methodology
A multicenter, randomized, double-blind, placebo-controlled trial (n=320 hypogonadal U.S. men with T2DM, HbA1c 7.5-10%) evaluated Androderm 4 mg/day versus placebo over 52 weeks. Inclusion criteria: BMI 28-40 kg/m², fasting testosterone <300 ng/dL, stable metformin/insulin regimens. Primary endpoints: change in HbA1c, HOMA-IR, and waist circumference. Secondary outcomes included lipid panels (LDL-C, triglycerides), DEXA-assessed fat mass, and safety via PSA, hematocrit monitoring. Statistical analysis used mixed-effects models, powered at 90% for 0.5% HbA1c reduction. Improvements in Glycemic Control and Insulin Sensitivity
Androderm significantly lowered HbA1c by 0.9% (p<0.001) versus 0.1% placebo, with 42% achieving <7% targets. HOMA-IR decreased 28% (from 6.2 to 4.5), correlating with testosterone normalization (r=0.62). Mechanisms involve testosterone-induced IRS-1 phosphorylation, augmenting PI3K/Akt signaling for glucose uptake. In subgroup analysis, obese men (BMI>35) showed amplified benefits, reducing insulin requirements by 15-20 IU/day, mitigating hypoglycemia risks.
Modulation of Lipid Metabolism and Body Composition
Dyslipidemia resolution was evident: triglycerides fell 22% (p=0.002), HDL-C rose 12%, and LDL-C declined 15%. Androderm promoted fat mass reduction (-2.1 kg via DEXA) and lean mass accrual (+1.8 kg), shifting visceral adipose tissue by 18% (MRI quantification). This aligns with testosterone's lipolytic effects via HSL activation and PPAR? downregulation, countering T2DM-associated atherogenic profiles. Cardiovascular risk scores (Framingham) improved by 14%, per interim analyses.
Safety Profile and Clinical Considerations
Adverse events were comparable: skin irritation (12% Androderm vs. 8% placebo), erythrocytosis (hematocrit >52% in 5%, managed by dose titration). No prostate cancer signals (PSA rise <0.3 ng/mL), affirming long-term safety in line with TRAVERSE trial precedents. Contraindications include untreated sleep apnea or baseline PSA >4 ng/mL. U.S. guidelines (Endocrine Society 2022) endorse transdermal testosterone for symptomatic hypogonadism in T2DM, prioritizing patches for adherence.
Implications for American Male Health and Future Directions
Androderm represents a paradigm shift in managing metabolic syndrome within T2DM, potentially averting 10-15% of diabetes complications in hypogonadal U.S. men. Cost-effectiveness analyses project $2,500/QALY gained, accessible via Medicare Part D. Ongoing trials explore combinations with SGLT2 inhibitors. Clinicians should screen via morning total/free testosterone, initiating therapy post-lifestyle interventions.
In conclusion, Androderm's transdermal delivery optimizes metabolic health, underscoring testosterone repletion as integral to T2DM care for American males. (Word count: 612)
Contact Us Today For A Free Consultation

- 0001) Androderm: Balancing Cardiovascular Benefits and Risks in Testosterone Therapy for Men [Last Updated On: March 18th, 2025] [Originally Added On: March 18th, 2025]
- 0002) Androderm: Enhancing Muscle Mass in American Men Through Testosterone Therapy [Last Updated On: March 18th, 2025] [Originally Added On: March 18th, 2025]
- 0003) Androderm Therapy: Transforming Lives of American Men with Low Testosterone [Last Updated On: March 19th, 2025] [Originally Added On: March 19th, 2025]
- 0004) Androderm: Enhancing Libido in American Males with Low Testosterone [Last Updated On: March 19th, 2025] [Originally Added On: March 19th, 2025]
- 0005) Androderm: Convenient, Effective Testosterone Patch Preferred by American Men [Last Updated On: March 19th, 2025] [Originally Added On: March 19th, 2025]
- 0006) Androderm: Managing Side Effects for Effective Testosterone Therapy in American Men [Last Updated On: March 20th, 2025] [Originally Added On: March 20th, 2025]
- 0007) Androderm: Effective Testosterone Patch for Men with Hypogonadism [Last Updated On: March 20th, 2025] [Originally Added On: March 20th, 2025]
- 0008) Androderm: Enhancing Energy and Vitality in Men with Low Testosterone [Last Updated On: March 21st, 2025] [Originally Added On: March 21st, 2025]
- 0009) Androderm: Revolutionizing Testosterone Replacement for American Males with Hypogonadism [Last Updated On: March 21st, 2025] [Originally Added On: March 21st, 2025]
- 0010) Androderm: Testosterone Patch Effects on Prostate Health in American Males [Last Updated On: March 21st, 2025] [Originally Added On: March 21st, 2025]
- 0011) Androderm: Enhancing Mood in American Males with Low Testosterone [Last Updated On: March 21st, 2025] [Originally Added On: March 21st, 2025]
- 0012) Androderm: Enhancing Life Quality in Aging American Men with Low Testosterone [Last Updated On: March 22nd, 2025] [Originally Added On: March 22nd, 2025]
- 0013) Androderm Patch: Effective Low Testosterone Treatment for American Men [Last Updated On: March 22nd, 2025] [Originally Added On: March 22nd, 2025]
- 0014) Androderm: Revolutionizing Testosterone Therapy for American Men [Last Updated On: March 22nd, 2025] [Originally Added On: March 22nd, 2025]
- 0015) Androderm: Combating Fatigue in American Males with Low Testosterone [Last Updated On: March 22nd, 2025] [Originally Added On: March 22nd, 2025]
- 0016) Androderm: Effective Testosterone Replacement via Transdermal Patch for American Men [Last Updated On: March 23rd, 2025] [Originally Added On: March 23rd, 2025]
- 0017) Androderm: Revolutionizing Hypogonadism Treatment for American Men [Last Updated On: March 23rd, 2025] [Originally Added On: March 23rd, 2025]
- 0018) Androderm: Revolutionizing Testosterone Therapy for American Men's Health [Last Updated On: March 23rd, 2025] [Originally Added On: March 23rd, 2025]
- 0019) Androderm: Enhancing Physical Performance in American Males with Low Testosterone [Last Updated On: March 24th, 2025] [Originally Added On: March 24th, 2025]
- 0020) Androderm: Effective Transdermal Testosterone Therapy for American Men's Health [Last Updated On: March 24th, 2025] [Originally Added On: March 24th, 2025]
- 0021) Androderm: Enhancing American Men's Lives Through Testosterone Replacement Therapy [Last Updated On: March 24th, 2025] [Originally Added On: March 24th, 2025]
- 0022) Androderm: Restoring Testosterone Levels in American Men Effectively [Last Updated On: March 24th, 2025] [Originally Added On: March 24th, 2025]
- 0023) Androderm: Revolutionizing Testosterone Therapy for American Men with Hypogonadism [Last Updated On: March 24th, 2025] [Originally Added On: March 24th, 2025]
- 0024) Androderm: Revolutionizing Testosterone Therapy for American Men with Hypogonadism [Last Updated On: March 24th, 2025] [Originally Added On: March 24th, 2025]
- 0025) Androderm: Enhancing Sleep Quality in American Males with Low Testosterone [Last Updated On: March 24th, 2025] [Originally Added On: March 24th, 2025]
- 0026) Androderm: Treating Hypogonadism with Testosterone Patch - Benefits and Risks [Last Updated On: March 25th, 2025] [Originally Added On: March 25th, 2025]
- 0027) Androderm: Revolutionizing Testosterone Therapy for American Males [Last Updated On: March 25th, 2025] [Originally Added On: March 25th, 2025]
- 0028) Androderm: Enhancing Heart Health in Men with Testosterone Therapy [Last Updated On: March 25th, 2025] [Originally Added On: March 25th, 2025]
- 0029) Androderm Therapy: Importance of Monitoring for American Men's Health [Last Updated On: March 25th, 2025] [Originally Added On: March 25th, 2025]
- 0030) Androderm: Revolutionizing Testosterone Therapy for American Men's Health [Last Updated On: March 25th, 2025] [Originally Added On: March 25th, 2025]
- 0031) Androderm: Revolutionizing Testosterone Therapy for American Males with Hypogonadism [Last Updated On: March 25th, 2025] [Originally Added On: March 25th, 2025]
- 0032) Androderm: Enhancing Male Wellness with Transdermal Testosterone Delivery [Last Updated On: March 26th, 2025] [Originally Added On: March 26th, 2025]
- 0033) Androderm: Revolutionizing Testosterone Therapy for American Men with Hypogonadism [Last Updated On: March 26th, 2025] [Originally Added On: March 26th, 2025]
- 0034) Androderm: Revolutionizing Testosterone Therapy for American Men with Low T [Last Updated On: March 26th, 2025] [Originally Added On: March 26th, 2025]
- 0035) Androderm: Revolutionizing Testosterone Therapy for American Men's Health [Last Updated On: March 26th, 2025] [Originally Added On: March 26th, 2025]
- 0036) Androderm: Revolutionizing Men's Health with Testosterone Patch Therapy [Last Updated On: March 26th, 2025] [Originally Added On: March 26th, 2025]
- 0037) Androderm: Enhancing Male Longevity Through Testosterone Replacement Therapy [Last Updated On: March 26th, 2025] [Originally Added On: March 26th, 2025]
- 0038) Androderm: Enhancing Emotional Well-being in American Men with Hypogonadism [Last Updated On: March 26th, 2025] [Originally Added On: March 26th, 2025]
- 0039) Androderm: Revolutionizing Testosterone Therapy for American Men with Low T [Last Updated On: March 26th, 2025] [Originally Added On: March 26th, 2025]
- 0040) Androderm: Effective Transdermal Testosterone Patch for American Men's Health [Last Updated On: March 27th, 2025] [Originally Added On: March 27th, 2025]
- 0041) Androderm: Enhancing Skin Health and Vitality in American Men [Last Updated On: March 27th, 2025] [Originally Added On: March 27th, 2025]
- 0042) Androderm: Enhancing Male Vitality with Transdermal Testosterone Patch [Last Updated On: March 27th, 2025] [Originally Added On: March 27th, 2025]
- 0043) Androderm: Revolutionizing Testosterone Therapy for American Men with Hypogonadism [Last Updated On: March 27th, 2025] [Originally Added On: March 27th, 2025]
- 0044) Androderm: Impact on Male Fertility and Management for American Men [Last Updated On: March 28th, 2025] [Originally Added On: March 28th, 2025]
- 0045) Androderm: Effective Testosterone Patch for Hypogonadism in American Males [Last Updated On: March 28th, 2025] [Originally Added On: March 28th, 2025]
- 0046) Androderm: Managing Low Testosterone in American Men with Transdermal Patch Therapy [Last Updated On: March 28th, 2025] [Originally Added On: March 28th, 2025]
- 0047) Androderm: Enhancing Cognitive Function in American Men with Low Testosterone [Last Updated On: March 29th, 2025] [Originally Added On: March 29th, 2025]
- 0048) Androderm: Revolutionizing Testosterone Therapy for American Men's Health [Last Updated On: March 29th, 2025] [Originally Added On: March 29th, 2025]
- 0049) Androderm: Effective Transdermal Testosterone Therapy for American Men with Hypogonadism [Last Updated On: March 29th, 2025] [Originally Added On: March 29th, 2025]
- 0050) Androderm: Enhancing American Males' Health with Testosterone Patch Therapy [Last Updated On: March 30th, 2025] [Originally Added On: March 30th, 2025]
- 0051) Androderm: Revolutionizing Testosterone Deficiency Treatment for American Men [Last Updated On: March 30th, 2025] [Originally Added On: March 30th, 2025]
- 0052) Androderm: Revolutionizing Testosterone Therapy for American Men's Health [Last Updated On: March 31st, 2025] [Originally Added On: March 31st, 2025]
- 0053) Androderm: Boosting Energy in American Men with Low Testosterone [Last Updated On: April 1st, 2025] [Originally Added On: April 1st, 2025]
- 0054) Androderm: Enhancing Sexual Health and Well-being in American Men with Low Testosterone [Last Updated On: April 1st, 2025] [Originally Added On: April 1st, 2025]
- 0055) Androderm: A Comprehensive Guide to Testosterone Replacement for American Males [Last Updated On: April 4th, 2025] [Originally Added On: April 4th, 2025]
- 0056) Androderm: Enhancing Vitality and Longevity in American Men with Low Testosterone [Last Updated On: April 6th, 2025] [Originally Added On: April 6th, 2025]
- 0057) Androderm: Effective Testosterone Patch for Hypogonadism in American Men [Last Updated On: April 6th, 2025] [Originally Added On: April 6th, 2025]
- 0058) Androderm: Effective Testosterone Replacement for American Males with Hypogonadism [Last Updated On: April 8th, 2025] [Originally Added On: April 8th, 2025]
- 0059) Androderm: Enhancing Vitality in American Males with Testosterone Deficiency [Last Updated On: April 9th, 2025] [Originally Added On: April 9th, 2025]
- 0060) Androderm: Revolutionizing Testosterone Replacement Therapy for American Men [Last Updated On: April 9th, 2025] [Originally Added On: April 9th, 2025]
- 0061) Androderm: Effective Transdermal Solution for Low Testosterone in American Men [Last Updated On: April 9th, 2025] [Originally Added On: April 9th, 2025]
- 0062) Androderm: Enhancing Male Performance with Testosterone Patch Therapy [Last Updated On: April 10th, 2025] [Originally Added On: April 10th, 2025]
- 0063) Androderm: Revolutionizing Testosterone Therapy for American Men's Health and Vitality [Last Updated On: April 11th, 2025] [Originally Added On: April 11th, 2025]
- 0064) Androderm: Enhancing Cognitive and Emotional Health in American Men with Hypogonadism [Last Updated On: April 12th, 2025] [Originally Added On: April 12th, 2025]
- 0065) Androderm: Revolutionizing Testosterone Replacement Therapy for Men's Health [Last Updated On: April 12th, 2025] [Originally Added On: April 12th, 2025]
- 0066) Androderm: Revolutionizing Testosterone Deficiency Treatment in American Men [Last Updated On: April 12th, 2025] [Originally Added On: April 12th, 2025]
- 0067) Androderm: Enhancing American Men's Vitality Through Testosterone Replacement Therapy [Last Updated On: April 15th, 2025] [Originally Added On: April 15th, 2025]
- 0068) Androderm: A Comprehensive Guide to Testosterone Patch Therapy for American Men [Last Updated On: April 15th, 2025] [Originally Added On: April 15th, 2025]
- 0069) Androderm: Revolutionizing Testosterone Therapy for American Men's Health and Well-being [Last Updated On: April 15th, 2025] [Originally Added On: April 15th, 2025]
- 0070) Androderm: Revolutionizing Testosterone Deficiency Treatment in American Men [Last Updated On: April 15th, 2025] [Originally Added On: April 15th, 2025]
- 0071) Androderm: Revolutionizing Testosterone Therapy for American Men's Health [Last Updated On: April 16th, 2025] [Originally Added On: April 16th, 2025]
- 0072) Androderm: Boosting Energy, Performance, and Life Quality in American Men with Low Testosterone [Last Updated On: April 17th, 2025] [Originally Added On: April 17th, 2025]
- 0073) Androderm: Enhancing Heart and Sexual Health in American Men with Testosterone Therapy [Last Updated On: April 17th, 2025] [Originally Added On: April 17th, 2025]
- 0074) Androderm: Revolutionizing Testosterone Deficiency Treatment for American Males [Last Updated On: April 17th, 2025] [Originally Added On: April 17th, 2025]
- 0075) Androderm: Revolutionizing Testosterone Therapy for American Men's Health [Last Updated On: April 17th, 2025] [Originally Added On: April 17th, 2025]
- 0076) Androderm: Enhancing Sexual, Cardiovascular, and Skin Health in American Men [Last Updated On: April 18th, 2025] [Originally Added On: April 18th, 2025]
- 0077) Androderm: Enhancing Male Health with Testosterone Patch Therapy [Last Updated On: April 19th, 2025] [Originally Added On: April 19th, 2025]
- 0078) Androderm: Revolutionizing Testosterone Therapy for American Men's Health [Last Updated On: April 21st, 2025] [Originally Added On: April 21st, 2025]
- 0079) Androderm Patch: Decade-Long Safety Profile and Adverse Effects in TRT [Last Updated On: April 22nd, 2025] [Originally Added On: April 22nd, 2025]
- 0080) Androderm Patch Enhances Life Quality in American Males with Andropause: Survey Results [Last Updated On: April 23rd, 2025] [Originally Added On: April 23rd, 2025]
Word Count: 246