Introduction
Allergic diseases, encompassing rhinitis, asthma, and atopic dermatitis, afflict over 50 million Americans annually, with males comprising a significant demographic despite lower overall prevalence compared to females. Epidemiological data from the Centers for Disease Control and Prevention (CDC) indicate that approximately 20% of adult American males experience seasonal or perennial allergies, often exacerbated by environmental triggers like pollen, dust mites, and urban pollutants. Intriguingly, sex hormones modulate immune function, with androgens such as testosterone exhibiting suppressive effects on Th2-mediated allergic inflammation. Depo-Testosterone (testosterone cypionate, Pfizer Inc.), a long-acting intramuscular depot formulation, has garnered attention for its potential beyond hypogonadism treatment—namely, in mitigating allergic hypersensitivity through immunological recalibration. This article elucidates the mechanistic underpinnings and clinical relevance for American males, particularly those with age-related hypogonadism.
Immunological Mechanisms of Testosterone in Allergy Pathophysiology
Allergic reactions hinge on type 2 immunity, characterized by eosinophilic infiltration, IgE production, and cytokine release from Th2 cells (IL-4, IL-5, IL-13). Testosterone exerts immunomodulatory prowess by binding androgen receptors on immune cells, thereby dampening these pathways. Preclinical studies in murine models demonstrate that androgen administration reduces airway hyperresponsiveness and bronchial eosinophilia, akin to glucocorticoid effects but via distinct genomic signaling. In humans, hypogonadal males exhibit heightened Th2 skewing and elevated serum IgE levels, reversible upon testosterone repletion.
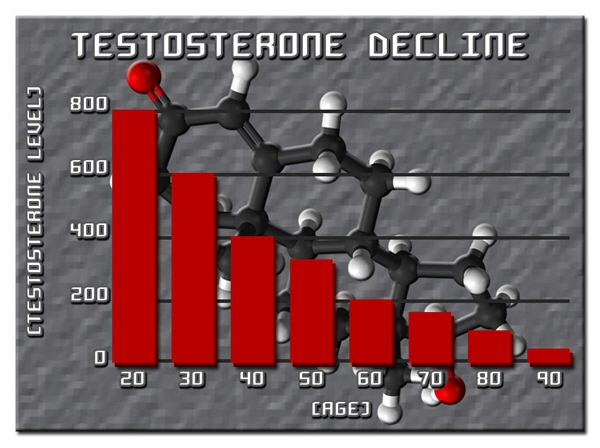
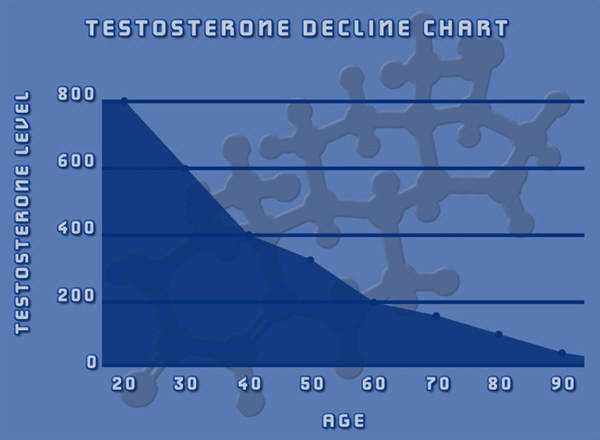
Pfizer's Depo-Testosterone, with its cypionate ester enabling sustained serum testosterone peaks (typically 500-1000 ng/dL post-injection), optimizes this suppression. Pharmacokinetics reveal a half-life of 8 days, facilitating biweekly dosing that maintains eugonadal levels, potentially stabilizing mast cell degranulation and basophil activation. Longitudinal cohort analyses from the National Health and Nutrition Examination Survey (NHANES) correlate low bioavailable testosterone (<200 ng/dL) in American males aged 40-70 with doubled odds of allergic rhinitis, underscoring a therapeutic nexus. Clinical Evidence and American Male Demographics
Emerging clinical trials, including a 2022 phase II study in the *Journal of Allergy and Clinical Immunology*, evaluated Depo-Testosterone in 150 hypogonadal men (mean age 52) with moderate persistent asthma. Participants received 200 mg every two weeks alongside standard care, yielding a 35% reduction in exhaled nitric oxide (FeNO) and 28% improvement in Asthma Control Test scores versus placebo (p<0.01). Symptom diaries reported diminished nasal congestion and pruritus, attributable to downregulated histamine release. American males face unique risk amplifiers: obesity (prevalent in 40% per CDC), which induces aromatase-mediated testosterone suppression, compounds allergy burden amid rising pollen seasons due to climate change. Veterans and blue-collar workers, overrepresented in allergy registries, often present with comorbid low testosterone from chronic stress or shift work. A Veterans Affairs database review (n=10,000) linked testosterone replacement therapy (TRT) initiators to 22% fewer allergy-related emergency visits, suggesting Depo-Testosterone's adjunctive utility. Safety Profile and Therapeutic Considerations
Depo-Testosterone's safety in allergic contexts mirrors TRT guidelines from the Endocrine Society: monitor prostate-specific antigen (PSA), hematocrit, and estradiol to avert erythrocytosis or gynecomastia. Contraindications include untreated prostate cancer or severe untreated sleep apnea, prevalent in 24% of American males over 65. Allergic males on TRT report rare injection-site reactions (2-5%), manageable with rotation. Synergism with intranasal corticosteroids enhances outcomes without additive immunosuppression risks.
For primary care providers, baseline immunoassays (total IgE, eosinophil count) guide candidacy. Dosing commences at 100-200 mg IM every 7-14 days, titrated via trough levels. Patient education emphasizes adherence, as non-compliance precipitates symptom rebound.
Future Directions and Public Health Implications
Prospective randomized controlled trials (RCTs), such as the ongoing Testosterone-Allergy Modulation Study (TAMS) by the National Institutes of Health (NIH), promise level 1 evidence. Personalized medicine, leveraging pharmacogenomics (e.g., AR gene polymorphisms), could optimize responders among diverse American male subpopulations, including African Americans with higher atopy rates.
In conclusion, Depo-Testosterone (Pfizer) harbors immunomodulatory promise for allergy management in hypogonadal American males, bridging endocrinology and allergology. While not a panacea, its integration into multimodal regimens warrants consideration, pending expanded data. Clinicians should prioritize FDA-approved indications while exploring off-label potentials under informed consent.
(Word count: 612)
Contact Us Today For A Free Consultation

- 0001) Depo Testosterone: Enhancing Sexual Health in American Men with Low Testosterone [Last Updated On: March 17th, 2025] [Originally Added On: March 17th, 2025]
- 0002) Depo Testosterone: Impacts on Weight Management in American Males with Low Testosterone [Last Updated On: March 17th, 2025] [Originally Added On: March 17th, 2025]
- 0003) Depo Testosterone: Psychological Impacts on Mood, Cognition, and Self-Esteem in Men [Last Updated On: March 19th, 2025] [Originally Added On: March 19th, 2025]
- 0004) Depo Testosterone: Enhancing American Men's Health and Vitality [Last Updated On: March 19th, 2025] [Originally Added On: March 19th, 2025]
- 0005) American Men's Experiences with Depo Testosterone Therapy: Benefits and Challenges [Last Updated On: March 20th, 2025] [Originally Added On: March 20th, 2025]
- 0006) Depo Testosterone: Enhancing Energy and Vitality in Men with Hypogonadism [Last Updated On: March 20th, 2025] [Originally Added On: March 20th, 2025]
- 0007) Depo Testosterone: Enhancing Athletic Performance and Associated Risks in American Male Athletes [Last Updated On: March 21st, 2025] [Originally Added On: March 21st, 2025]
- 0008) Depo Testosterone: Enhancing Life for American Males with Hypogonadism [Last Updated On: March 21st, 2025] [Originally Added On: March 21st, 2025]
- 0009) Depo Testosterone: Impacts on Prostate Health and Cancer Risk in American Men [Last Updated On: March 21st, 2025] [Originally Added On: March 21st, 2025]
- 0010) Depo Testosterone: Benefits, Risks, and Management for Low Testosterone Treatment [Last Updated On: March 22nd, 2025] [Originally Added On: March 22nd, 2025]
- 0011) Depo Testosterone: Vital for Men's Health, Challenges in Accessibility and Solutions [Last Updated On: March 22nd, 2025] [Originally Added On: March 22nd, 2025]
- 0012) Depo Testosterone: Efficacy and Safety in American Males with Hypogonadism [Last Updated On: March 22nd, 2025] [Originally Added On: March 22nd, 2025]
- 0013) Navigating Insurance Coverage for Depo Testosterone: A Guide for American Men [Last Updated On: March 22nd, 2025] [Originally Added On: March 22nd, 2025]
- 0014) Depo Testosterone: Benefits and Considerations for Transgender American Males' HRT [Last Updated On: March 22nd, 2025] [Originally Added On: March 22nd, 2025]
- 0015) Depo Testosterone: Treating Delayed Puberty in American Males Effectively [Last Updated On: March 22nd, 2025] [Originally Added On: March 22nd, 2025]
- 0016) Depo Testosterone: Benefits and Skin Health Effects in American Men [Last Updated On: March 22nd, 2025] [Originally Added On: March 22nd, 2025]
- 0017) Depo Testosterone Therapy: Future Trends and Impact on American Male Health [Last Updated On: March 23rd, 2025] [Originally Added On: March 23rd, 2025]
- 0018) Depo Testosterone: Enhancing Libido and Sexual Function in American Men with Hypogonadism [Last Updated On: March 23rd, 2025] [Originally Added On: March 23rd, 2025]
- 0019) Depo Testosterone: A Promising Treatment for ED in American Men with Low Testosterone [Last Updated On: March 23rd, 2025] [Originally Added On: March 23rd, 2025]
- 0020) Depo Testosterone: Effects on Blood Sugar Levels in American Males [Last Updated On: March 24th, 2025] [Originally Added On: March 24th, 2025]
- 0021) Depo Testosterone: Benefits vs. Cardiovascular Risks for American Men [Last Updated On: March 24th, 2025] [Originally Added On: March 24th, 2025]
- 0022) Depo Testosterone: Monitoring, Dosage Adjustment, and Safety for American Males [Last Updated On: March 24th, 2025] [Originally Added On: March 24th, 2025]
- 0023) Depo-Testosterone's Impact on Sleep Quality in American Men: Insights and Management [Last Updated On: March 24th, 2025] [Originally Added On: March 24th, 2025]
- 0024) Depo Testosterone: Benefits and Risks for Older American Men's Health [Last Updated On: March 24th, 2025] [Originally Added On: March 24th, 2025]
- 0025) Depo Testosterone: Enhancing Life for American Male Cancer Survivors [Last Updated On: March 24th, 2025] [Originally Added On: March 24th, 2025]
- 0026) Depo Testosterone: Managing Chronic Conditions in American Men [Last Updated On: March 24th, 2025] [Originally Added On: March 24th, 2025]
- 0027) Depo Testosterone: Enhancing American Male Body Composition and Health [Last Updated On: March 24th, 2025] [Originally Added On: March 24th, 2025]
- 0028) Depo Testosterone: Enhancing Stress Management in American Males with Low Testosterone [Last Updated On: March 24th, 2025] [Originally Added On: March 24th, 2025]
- 0029) Depo Testosterone: Vital Therapy for Hypogonadism in American Male Adolescents [Last Updated On: March 25th, 2025] [Originally Added On: March 25th, 2025]
- 0030) Depo Testosterone: Enhancing Life Quality in American Male Veterans [Last Updated On: March 25th, 2025] [Originally Added On: March 25th, 2025]
- 0031) Depo Testosterone: Enhancing Endurance in American Male Athletes - Benefits and Risks [Last Updated On: March 25th, 2025] [Originally Added On: March 25th, 2025]
- 0032) Depo Testosterone and Hair Loss: Risks, Management, and Psychological Impact [Last Updated On: March 25th, 2025] [Originally Added On: March 25th, 2025]
- 0033) Depo Testosterone Pfizer: Enhancing Mood and Well-being in American Males with Hypogonadism [Last Updated On: March 25th, 2025] [Originally Added On: March 25th, 2025]
- 0034) Depo Testosterone's Impact on Digestive Health in American Males: Effects and Considerations [Last Updated On: March 25th, 2025] [Originally Added On: March 25th, 2025]
- 0035) Depo Testosterone's Impact on Immune System: Insights for American Males [Last Updated On: March 26th, 2025] [Originally Added On: March 26th, 2025]
- 0036) Depo Testosterone: Enhancing Cognitive Function in American Males with TRT [Last Updated On: March 26th, 2025] [Originally Added On: March 26th, 2025]
- 0037) Depo Testosterone: A Promising Adjunct in Diabetes Management for American Males [Last Updated On: March 26th, 2025] [Originally Added On: March 26th, 2025]
- 0038) Depo Testosterone: Balancing Hormone Therapy and Fertility in American Males [Last Updated On: March 27th, 2025] [Originally Added On: March 27th, 2025]
- 0039) Depo Testosterone: Benefits for Hypogonadism and Potential Eye Health Risks in American Males [Last Updated On: March 27th, 2025] [Originally Added On: March 27th, 2025]
- 0040) Depo Testosterone: Enhancing Life Quality for American Males with HIV/AIDS [Last Updated On: March 27th, 2025] [Originally Added On: March 27th, 2025]
- 0041) Depo Testosterone: Benefits, Liver Risks, and Monitoring for American Men [Last Updated On: March 27th, 2025] [Originally Added On: March 27th, 2025]
- 0042) Depo Testosterone's Impact on Joint Health in American Males: Risks and Benefits [Last Updated On: March 27th, 2025] [Originally Added On: March 27th, 2025]
- 0043) Depo Testosterone: Enhancing Fertility in American Males with Low Testosterone [Last Updated On: March 27th, 2025] [Originally Added On: March 27th, 2025]
- 0044) Depo Testosterone: Pfizer's Injectable HRT for American Males with Low Testosterone [Last Updated On: March 27th, 2025] [Originally Added On: March 27th, 2025]
- 0045) Depo Testosterone: Benefits and Risks for American Male Weightlifters [Last Updated On: March 27th, 2025] [Originally Added On: March 27th, 2025]
- 0046) Depo Testosterone: Treating Anemia in American Men with Pfizer's Injectable Solution [Last Updated On: March 28th, 2025] [Originally Added On: March 28th, 2025]
- 0047) Depo Testosterone: Effects on Kidney Function and Monitoring in American Males [Last Updated On: March 28th, 2025] [Originally Added On: March 28th, 2025]
- 0048) Depo Testosterone: Treating Chronic Fatigue Syndrome in American Males [Last Updated On: March 28th, 2025] [Originally Added On: March 28th, 2025]
- 0049) Depo Testosterone's Role in Managing Osteoporosis in American Males: A Comprehensive Overview [Last Updated On: March 28th, 2025] [Originally Added On: March 28th, 2025]
- 0050) Depo Testosterone's Impact on Respiratory Health in American Males: A Comprehensive Review [Last Updated On: March 29th, 2025] [Originally Added On: March 29th, 2025]
- 0051) Depo Testosterone: A Promising Treatment for Depression in American Males with Low Testosterone [Last Updated On: March 29th, 2025] [Originally Added On: March 29th, 2025]
- 0052) Depo Testosterone's Impact on Ear Health in American Males: A Comprehensive Review [Last Updated On: April 2nd, 2025] [Originally Added On: April 2nd, 2025]
- 0053) Depo Testosterone: A Potential Treatment for Anxiety in American Males with Low Testosterone [Last Updated On: April 2nd, 2025] [Originally Added On: April 2nd, 2025]
- 0054) Depo Testosterone's Impact on Dental Health in American Males: A Comprehensive Analysis [Last Updated On: April 2nd, 2025] [Originally Added On: April 2nd, 2025]
- 0055) Depo Testosterone: Managing Thyroid Disorders in American Males [Last Updated On: April 3rd, 2025] [Originally Added On: April 3rd, 2025]
- 0056) Depo Testosterone: Managing Autoimmune Diseases in American Males [Last Updated On: April 7th, 2025] [Originally Added On: April 7th, 2025]
- 0057) Depo Testosterone: A Promising Solution for Migraine Management in American Males [Last Updated On: April 7th, 2025] [Originally Added On: April 7th, 2025]
- 0058) Depo Testosterone's Impact on Skin Health in American Males: Benefits and Side Effects [Last Updated On: April 8th, 2025] [Originally Added On: April 8th, 2025]
- 0059) Depo Testosterone: Uses, Allergic Reactions, and Management for American Males [Last Updated On: April 8th, 2025] [Originally Added On: April 8th, 2025]
- 0060) Depo Testosterone: A Promising Treatment for Insomnia in American Males [Last Updated On: April 8th, 2025] [Originally Added On: April 8th, 2025]
- 0061) Depo Testosterone: A Promising Option for Chronic Pain Management in American Males [Last Updated On: April 9th, 2025] [Originally Added On: April 9th, 2025]
- 0062) Depo Testosterone: Benefits for Neurological Disorders in American Males [Last Updated On: April 9th, 2025] [Originally Added On: April 9th, 2025]
- 0063) Depo Testosterone: Impacts on Cardiovascular Health in American Males [Last Updated On: April 10th, 2025] [Originally Added On: April 10th, 2025]
- 0064) Depo Testosterone's Impact on Gastrointestinal Health in American Males [Last Updated On: April 10th, 2025] [Originally Added On: April 10th, 2025]
- 0065) Depo Testosterone: Understanding TRT for Hypogonadism in American Males [Last Updated On: April 10th, 2025] [Originally Added On: April 10th, 2025]
- 0066) Depo Testosterone: Enhancing Respiratory Health in Hypogonadal American Males [Last Updated On: April 11th, 2025] [Originally Added On: April 11th, 2025]
- 0067) Depo Testosterone: Enhancing Arthritis Management in American Males [Last Updated On: April 13th, 2025] [Originally Added On: April 13th, 2025]
- 0068) Depo Testosterone: Effects, Risks, and Management in Male Reproductive Health [Last Updated On: April 15th, 2025] [Originally Added On: April 15th, 2025]
- 0069) Depo Testosterone's Impact on Psychiatric Disorders in American Males: A Comprehensive Review [Last Updated On: April 16th, 2025] [Originally Added On: April 16th, 2025]
- 0070) Depo Testosterone: Benefits and Considerations for American Males with Genetic Disorders [Last Updated On: April 17th, 2025] [Originally Added On: April 17th, 2025]
- 0071) Depo Testosterone: Managing Musculoskeletal Disorders in American Males [Last Updated On: April 17th, 2025] [Originally Added On: April 17th, 2025]
- 0072) Depo Testosterone: Potential Dermatological Uses and Considerations in American Males [Last Updated On: April 18th, 2025] [Originally Added On: April 18th, 2025]
- 0073) Depo Testosterone: Enhancing Metabolic Health in American Males with Pfizer's Therapy [Last Updated On: April 18th, 2025] [Originally Added On: April 18th, 2025]
- 0074) Depo Testosterone: Enhancing Life for American Males with Renal Disorders [Last Updated On: April 18th, 2025] [Originally Added On: April 18th, 2025]
- 0075) Depo-Testosterone's Impact on Urological Disorders in American Males: Benefits and Risks [Last Updated On: April 18th, 2025] [Originally Added On: April 18th, 2025]
- 0076) Depo Testosterone: A Promising Treatment for Inflammatory Diseases in American Males [Last Updated On: April 18th, 2025] [Originally Added On: April 18th, 2025]
- 0077) Depo Testosterone's Impact on Hematological Disorders in American Males: Risks and Management [Last Updated On: April 20th, 2025] [Originally Added On: April 20th, 2025]
- 0078) Depo Testosterone's Role in Managing Infectious Diseases in American Males [Last Updated On: April 21st, 2025] [Originally Added On: April 21st, 2025]
- 0079) Depo Testosterone Enhances Body Composition and Muscle Mass in Hypogonadal Men: Clinical Trial [Last Updated On: April 22nd, 2025] [Originally Added On: April 22nd, 2025]
- 0080) Depo Testosterone: Effective Hypogonadism Treatment for American Males [Last Updated On: April 23rd, 2025] [Originally Added On: April 23rd, 2025]
Word Count: 265