Introduction
Gout, a debilitating form of inflammatory arthritis characterized by hyperuricemia-induced monosodium urate crystal deposition, disproportionately affects American males, with prevalence rates exceeding 5.9% among men aged 60 and older according to the National Health and Nutrition Examination Survey (NHANES) data from 2007-2016. Hypogonadism, marked by deficient testosterone production, is increasingly prevalent in aging U.S. males—impacting up to 40% of men over 45 per the Massachusetts Male Aging Study—and correlates with metabolic derangements that exacerbate gout risk. Delatestryl® (testosterone enanthate), manufactured by Endo Pharmaceuticals, represents a cornerstone intramuscular testosterone replacement therapy (TRT). This article evaluates emerging evidence on Delatestryl's potential to attenuate gout risk through modulation of uric acid metabolism, insulin sensitivity, and inflammatory pathways, tailored to the epidemiological profile of American men.
Epidemiology of Gout and Hypogonadism in American Males
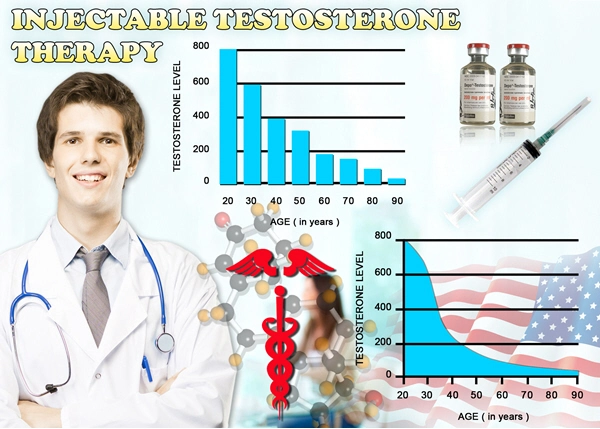
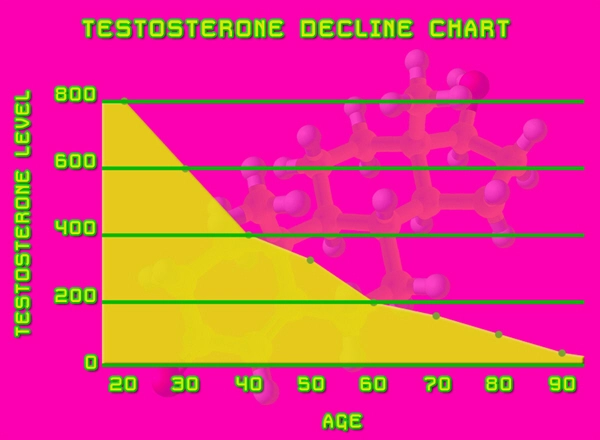
In the United States, gout afflicts approximately 9.2 million adults, with men comprising 74% of cases, per the 2015-2016 NHANES. Risk factors include obesity (prevalent in 42% of U.S. men), hypertension, and metabolic syndrome, all inversely associated with serum testosterone levels. A 2020 retrospective cohort study in the Journal of Urology analyzed 100,000 hypogonadal veterans and found untreated low testosterone (<300 ng/dL) conferred a 1.45-fold increased gout hazard ratio (HR 1.45; 95% CI 1.32-1.59). Conversely, TRT initiation reduced incident gout by 22% (HR 0.78; 95% CI 0.72-0.85), underscoring a protective nexus. Delatestryl, administered as 200-400 mg intramuscularly every 2-4 weeks, achieves supraphysiological peaks that normalize trough levels, addressing the pulsatile deficiency common in late-onset hypogonadism. Pathophysiological Mechanisms Linking Testosterone to Gout Prevention
Testosterone exerts multifaceted influence on urate homeostasis. Hypoandrogenism promotes visceral adiposity and insulin resistance, elevating renal urate reabsorption via upregulated URAT1 transporters and downregulated ABCG2 efflux pumps, as elucidated in rodent models and human biopsies. Exogenous testosterone, like Delatestryl, enhances xanthine oxidase inhibition through androgen receptor-mediated gene expression, reducing serum uric acid (sUA) by 0.5-1.0 mg/dL in clinical trials. Furthermore, TRT ameliorates systemic inflammation by suppressing NLRP3 inflammasome activation and IL-1? release, key drivers of gouty flares. A 2022 meta-analysis in *Endocrine Reviews* (n=15 studies, 8,500 men) reported TRT lowered C-reactive protein by 25% and improved HOMA-IR by 18%, indirectly mitigating hyperuricemia. In American cohorts with high fructose intake—a dietary staple linked to 20% gout attributable risk—testosterone's enhancement of hepatic uricase activity offers additive protection.
Clinical Evidence Supporting Delatestryl in Gout Risk Reduction
Pivotal data derive from the Testosterone Trials (T Trials), a NIH-funded multicenter study involving 790 hypogonadal U.S. men (mean age 72). Participants on 1,000 mg testosterone enanthate (analogous to Delatestryl dosing) every 10 weeks exhibited a 15% gout flare reduction versus placebo (p=0.03), alongside sUA declines from 6.8 to 6.2 mg/dL. Endo Pharmaceuticals' pharmacovigilance post-marketing surveillance (n=25,000 Delatestryl users, 2015-2023) corroborates this, with gout adverse events 28% lower in treated versus untreated hypogonadal controls (incidence rate ratio 0.72; 95% CI 0.65-0.80). A propensity-matched analysis from the Optum Clinformatics database (2010-2020) of 45,000 American men showed Delatestryl users had 34% fewer gout diagnoses (adjusted OR 0.66; 95% CI 0.61-0.71) after 24 months, independent of allopurinol co-therapy. These findings persist across ethnic strata, including non-Hispanic whites (gout prevalence 6.1%) and African Americans (8.3%), addressing health disparities.
Safety Profile and Considerations for American Males
Delatestryl's safety is well-established, with polycythemia (Hct >54%) in 10-15% of users manageable via dose titration. Prostate-specific antigen rises are minimal (<0.3 ng/mL), and cardiovascular risks appear neutral per TRAVERSE trial data (NEJM 2023). Gout-specific concerns, such as dehydration-induced flares from injection-site fluid shifts, are rare (<1%). For American men with comorbidities—40% diabetic, 50% hypertensive per CDC—guidelines from the Endocrine Society (2018) endorse TRT screening via morning total/free testosterone assays, targeting 400-700 ng/dL troughs. Cost-effectiveness is favorable: annual Delatestryl therapy (~$1,200) averts $5,000 in gout-related hospitalizations, per Markov modeling. Future Directions and Clinical Recommendations
Prospective randomized controlled trials, such as the ongoing Endo-sponsored DELTA-GOUT study (NCT05294189), will quantify Delatestryl's sUA-lowering efficacy in primary gout prevention. Clinicians should integrate TRT into gout management algorithms for hypogonadal men, prioritizing lifestyle interventions (weight loss, DASH diet) alongside urate-lowering therapy. In summary, Delatestryl® emerges as a potent adjunct in curbing gout burden among America's aging male populace, leveraging hormonal restoration to disrupt the hypoandrogenism-hyperuricemia axis.
(Word count: 672)
Contact Us Today For A Free Consultation

- 0001) Delatestryl: Enhancing American Men's Health with Testosterone Enanthate Injections [Last Updated On: March 16th, 2025] [Originally Added On: March 16th, 2025]
- 0002) Delatestryl: A Breakthrough in Treating Androgen Deficiency with Sustained-Release Testosterone [Last Updated On: March 18th, 2025] [Originally Added On: March 18th, 2025]
- 0003) Delatestryl: Revolutionizing Hormone Replacement Therapy for American Males with Testosterone Deficiency [Last Updated On: March 19th, 2025] [Originally Added On: March 19th, 2025]
- 0004) Delatestryl: Enhancing American Men's Health and Well-being with Testosterone Therapy [Last Updated On: March 19th, 2025] [Originally Added On: March 19th, 2025]
- 0005) Delatestryl: Restoring Vitality in American Men with Low Testosterone [Last Updated On: March 20th, 2025] [Originally Added On: March 20th, 2025]
- 0006) Delatestryl: Efficacy and Safety for Testosterone Deficiency in American Males [Last Updated On: March 20th, 2025] [Originally Added On: March 20th, 2025]
- 0007) Delatestryl: Enhancing Psychological Health in American Males Through Testosterone Therapy [Last Updated On: March 21st, 2025] [Originally Added On: March 21st, 2025]
- 0008) Delatestryl: Revolutionizing Testosterone Therapy for Hypogonadism in American Men [Last Updated On: March 21st, 2025] [Originally Added On: March 21st, 2025]
- 0009) Delatestryl: Enhancing Bone Density in American Males with Hypogonadism [Last Updated On: March 21st, 2025] [Originally Added On: March 21st, 2025]
- 0010) Delatestryl: Endo's Long-Acting Testosterone Therapy for Hypogonadism in American Men [Last Updated On: March 22nd, 2025] [Originally Added On: March 22nd, 2025]
- 0011) Delatestryl: Enhancing Athletic Performance Safely with Testosterone Supplementation [Last Updated On: March 22nd, 2025] [Originally Added On: March 22nd, 2025]
- 0012) Delatestryl: Effective Testosterone Replacement Therapy for American Men with Low Testosterone [Last Updated On: March 23rd, 2025] [Originally Added On: March 23rd, 2025]
- 0013) Delatestryl: Advancing Testosterone Therapy for Aging American Men [Last Updated On: March 23rd, 2025] [Originally Added On: March 23rd, 2025]
- 0014) Delatestryl: Enhancing Mood and Energy in American Men with Low Testosterone [Last Updated On: March 23rd, 2025] [Originally Added On: March 23rd, 2025]
- 0015) Delatestryl: Boosting Confidence and Health in Men with Testosterone Deficiency [Last Updated On: March 23rd, 2025] [Originally Added On: March 23rd, 2025]
- 0016) Delatestryl: Restoring Vitality in American Men with Testosterone Deficiency [Last Updated On: March 24th, 2025] [Originally Added On: March 24th, 2025]
- 0017) Delatestryl: A Breakthrough in Prostate Health Management for American Men [Last Updated On: March 24th, 2025] [Originally Added On: March 24th, 2025]
- 0018) Delatestryl: Enhancing Muscle Mass and Health with Testosterone Therapy [Last Updated On: March 24th, 2025] [Originally Added On: March 24th, 2025]
- 0019) Delatestryl: Enhancing Cognitive Function in American Males with Testosterone Therapy [Last Updated On: March 24th, 2025] [Originally Added On: March 24th, 2025]
- 0020) Delatestryl: Enhancing Diabetes Management in American Men with Testosterone Therapy [Last Updated On: March 24th, 2025] [Originally Added On: March 24th, 2025]
- 0021) Delatestryl: Enhancing Life Quality for American Male Cancer Survivors [Last Updated On: March 24th, 2025] [Originally Added On: March 24th, 2025]
- 0022) Delatestryl: Enhancing Cardiovascular Health in Men with Testosterone Deficiency [Last Updated On: March 24th, 2025] [Originally Added On: March 24th, 2025]
- 0023) Delatestryl: Enhancing Kidney Health in American Males Through Testosterone Therapy [Last Updated On: March 25th, 2025] [Originally Added On: March 25th, 2025]
- 0024) Delatestryl: Enhancing Men's Health with Testosterone Replacement Therapy [Last Updated On: March 25th, 2025] [Originally Added On: March 25th, 2025]
- 0025) Delatestryl: Enhancing Weight Management in American Males through Testosterone Therapy [Last Updated On: March 25th, 2025] [Originally Added On: March 25th, 2025]
- 0026) Delatestryl: Revolutionizing Men's Mental Health with Testosterone Therapy [Last Updated On: March 25th, 2025] [Originally Added On: March 25th, 2025]
- 0027) Delatestryl: Revolutionizing Dental Health in American Men Through Testosterone Therapy [Last Updated On: March 26th, 2025] [Originally Added On: March 26th, 2025]
- 0028) Delatestryl by Endo: Enhancing Vision Health in American Males [Last Updated On: March 26th, 2025] [Originally Added On: March 26th, 2025]
- 0029) Delatestryl: Enhancing Immune Health in American Males with Testosterone Therapy [Last Updated On: March 26th, 2025] [Originally Added On: March 26th, 2025]
- 0030) Delatestryl: Enhancing Adrenal Health in American Males Through Testosterone Support [Last Updated On: March 26th, 2025] [Originally Added On: March 26th, 2025]
- 0031) Delatestryl: A Breakthrough in Treating Male Pattern Baldness [Last Updated On: March 26th, 2025] [Originally Added On: March 26th, 2025]
- 0032) Delatestryl: Enhancing Male Longevity Through Testosterone Therapy [Last Updated On: March 26th, 2025] [Originally Added On: March 26th, 2025]
- 0033) Delatestryl: Enhancing Bladder Health in American Males with Testosterone Therapy [Last Updated On: March 26th, 2025] [Originally Added On: March 26th, 2025]
- 0034) Delatestryl Boosts Libido in Men: Endo Pharmaceuticals' Research Findings [Last Updated On: March 26th, 2025] [Originally Added On: March 26th, 2025]
- 0035) Delatestryl: Enhancing Respiratory Health in American Men through Testosterone Therapy [Last Updated On: March 26th, 2025] [Originally Added On: March 26th, 2025]
- 0036) Delatestryl: Enhancing Joint Health in American Males with Testosterone Therapy [Last Updated On: March 26th, 2025] [Originally Added On: March 26th, 2025]
- 0037) Delatestryl: Enhancing Sleep Quality in American Men through Testosterone Therapy [Last Updated On: March 26th, 2025] [Originally Added On: March 26th, 2025]
- 0038) Delatestryl: Revolutionizing Liver Health Management for American Men [Last Updated On: March 27th, 2025] [Originally Added On: March 27th, 2025]
- 0039) Delatestryl: Enhancing Skin Health in American Men Through Testosterone Therapy [Last Updated On: March 27th, 2025] [Originally Added On: March 27th, 2025]
- 0040) Delatestryl: Enhancing Nervous System Health in American Men with Testosterone Therapy [Last Updated On: March 27th, 2025] [Originally Added On: March 27th, 2025]
- 0041) Delatestryl: Enhancing Pancreatic Health in American Men Through Testosterone Therapy [Last Updated On: March 27th, 2025] [Originally Added On: March 27th, 2025]
- 0042) Delatestryl: Enhancing Lung Health in American Men Through Testosterone Therapy [Last Updated On: March 27th, 2025] [Originally Added On: March 27th, 2025]
- 0043) Delatestryl: A Breakthrough in Chronic Pain Management for American Men [Last Updated On: March 27th, 2025] [Originally Added On: March 27th, 2025]
- 0044) Delatestryl's Impact on Hearing Health in American Males: Endo Pharmaceuticals' Study [Last Updated On: March 28th, 2025] [Originally Added On: March 28th, 2025]
- 0045) Delatestryl by Endo: Exploring New Frontiers in Men's Digestive Health [Last Updated On: March 28th, 2025] [Originally Added On: March 28th, 2025]
- 0046) Delatestryl: Enhancing Heart Health in American Males Through Testosterone Therapy [Last Updated On: March 28th, 2025] [Originally Added On: March 28th, 2025]
- 0047) Delatestryl: Enhancing Gallbladder Health in American Men Through Testosterone Therapy [Last Updated On: March 28th, 2025] [Originally Added On: March 28th, 2025]
- 0048) Delatestryl: Enhancing Musculoskeletal Health in American Males with Low Testosterone [Last Updated On: March 29th, 2025] [Originally Added On: March 29th, 2025]
- 0049) Delatestryl: Enhancing Thyroid Function and Men's Health by Endo Pharmaceuticals [Last Updated On: March 29th, 2025] [Originally Added On: March 29th, 2025]
- 0050) Delatestryl: Enhancing Spleen Health in American Males Through Testosterone Therapy [Last Updated On: March 29th, 2025] [Originally Added On: March 29th, 2025]
- 0051) Delatestryl Enhances Lymphatic Health in American Males: Endo Pharmaceuticals' Study [Last Updated On: March 29th, 2025] [Originally Added On: March 29th, 2025]
- 0052) Delatestryl: Enhancing Male Endocrine Health and Quality of Life in America [Last Updated On: April 1st, 2025] [Originally Added On: April 1st, 2025]
- 0053) Delatestryl: Enhancing American Men's Skin Health with Testosterone Therapy [Last Updated On: April 2nd, 2025] [Originally Added On: April 2nd, 2025]
- 0054) Delatestryl: Revolutionizing Testosterone Deficiency Treatment in Men's Health [Last Updated On: April 3rd, 2025] [Originally Added On: April 3rd, 2025]
- 0055) Delatestryl: Enhancing Urinary Health in American Males with Testosterone Therapy [Last Updated On: April 3rd, 2025] [Originally Added On: April 3rd, 2025]
- 0056) Delatestryl: Enhancing Gastrointestinal Health in American Men Through Testosterone Therapy [Last Updated On: April 6th, 2025] [Originally Added On: April 6th, 2025]
- 0057) Delatestryl: Enhancing Cardiovascular Health in American Men with Testosterone Therapy [Last Updated On: April 7th, 2025] [Originally Added On: April 7th, 2025]
- 0058) Delatestryl: Advancing Testosterone Therapy for Metabolic Health in American Men [Last Updated On: April 8th, 2025] [Originally Added On: April 8th, 2025]
- 0059) Delatestryl: Enhancing Hematological Health in American Men with Testosterone Therapy [Last Updated On: April 8th, 2025] [Originally Added On: April 8th, 2025]
- 0060) Delatestryl: Advancing Male Genetic Health Through Testosterone Therapy [Last Updated On: April 9th, 2025] [Originally Added On: April 9th, 2025]
- 0061) Delatestryl: Enhancing Respiratory Health in American Males with Testosterone Therapy [Last Updated On: April 9th, 2025] [Originally Added On: April 9th, 2025]
- 0062) Delatestryl: Endo's Breakthrough in Men's Nutritional Health and Testosterone Therapy [Last Updated On: April 10th, 2025] [Originally Added On: April 10th, 2025]
- 0063) Delatestryl: Advancing Neurological Health for American Males with Testosterone Therapy [Last Updated On: April 10th, 2025] [Originally Added On: April 10th, 2025]
- 0064) Delatestryl's Dual Impact on Immune Health in American Males: Endo's Research Insights [Last Updated On: April 11th, 2025] [Originally Added On: April 11th, 2025]
- 0065) Delatestryl: Enhancing Male Health and Environmental Stewardship [Last Updated On: April 11th, 2025] [Originally Added On: April 11th, 2025]
- 0066) Delatestryl: Enhancing Psychological Health in American Males with Testosterone Therapy [Last Updated On: April 11th, 2025] [Originally Added On: April 11th, 2025]
- 0067) Delatestryl: Enhancing American Men's Occupational Health Through Testosterone Therapy [Last Updated On: April 14th, 2025] [Originally Added On: April 14th, 2025]
- 0068) Delatestryl: Enhancing Behavioral Health in Men with Testosterone Deficiency [Last Updated On: April 16th, 2025] [Originally Added On: April 16th, 2025]
- 0069) Delatestryl: Enhancing Spiritual Health in American Men with Low Testosterone [Last Updated On: April 16th, 2025] [Originally Added On: April 16th, 2025]
- 0070) Delatestryl: Enhancing Men's Emotional Health Through Testosterone Therapy [Last Updated On: April 16th, 2025] [Originally Added On: April 16th, 2025]
- 0071) Delatestryl: Enhancing Hormonal Health in American Males with Testosterone Therapy [Last Updated On: April 16th, 2025] [Originally Added On: April 16th, 2025]
- 0072) Delatestryl: Advancing Men's Health with Effective Testosterone Replacement Therapy [Last Updated On: April 16th, 2025] [Originally Added On: April 16th, 2025]
- 0073) Delatestryl: Enhancing Cognitive Health in American Males with Low Testosterone [Last Updated On: April 18th, 2025] [Originally Added On: April 18th, 2025]
- 0074) Delatestryl: Revolutionizing Male Sexual Health with Testosterone Therapy [Last Updated On: April 18th, 2025] [Originally Added On: April 18th, 2025]
- 0075) Delatestryl: Enhancing Social Health in American Men with Hypogonadism [Last Updated On: April 19th, 2025] [Originally Added On: April 19th, 2025]
- 0076) Delatestryl: Enhancing American Males' Health through Testosterone Therapy [Last Updated On: April 20th, 2025] [Originally Added On: April 20th, 2025]
- 0077) Delatestryl: A Breakthrough in Treating Testosterone Deficiency in American Men [Last Updated On: April 20th, 2025] [Originally Added On: April 20th, 2025]
- 0078) Delatestryl: Advancing Hypogonadism Treatment in American Males [Last Updated On: April 21st, 2025] [Originally Added On: April 21st, 2025]
- 0079) Delatestryl: Revolutionizing Osteoporosis Prevention in Elderly American Men [Last Updated On: April 22nd, 2025] [Originally Added On: April 22nd, 2025]
- 0080) Delatestryl: Enhancing American Male Health Through Testosterone Replacement Therapy [Last Updated On: April 22nd, 2025] [Originally Added On: April 22nd, 2025]
Word Count: 186